Study on application of reduced Graphene Oxide on cotton fabric for Breathable Waterproof finish
Study to investigate the effects of waterproof finishes with breath-ability by using reduced rGO & PUA
This article explains the application of reduced graphene oxide on cotton woven fabric to achieve breathable waterproof finishing. After finishing the fabric was analysed various morphological properties as well as physical properties to confirm comfortability.
Abstract
In this study, 100% cotton woven fabric was chosen to investigate the effects of waterproof finishes with breathability by using reduced Graphene Oxide (rGO) and polyurethane binder (PUA). The effects of the waterproof of the fabrics were measured in accordance with AATCC 22 (1996) spray test and contact angle method. Evaluate the physical properties (tensile strength, stiffness test, thickness test) of waterproof finished fabric were evaluated by using relevant British Standards, ASTM, and ISO standards. The wetting time of the fabrics was also measured before and after treated samples to evaluate the performance and durability of the finishing.
Introduction
The onset of the 21st century has marked great developments in technology and science. However, these developments have come with a price, one of which is aggravated global warming, leading to sudden climatic changes. In order to survive and be protected in such conditions, there is a need for appropriate apparel and workwear for people specifically working in outdoor environments.
The study of graphene has been one of the most exciting topics in material science and many other research fields since the first report of the preparation and isolation of single graphene layers in 2004. Flexible textile functional materials have recently attracted the attention for their Antibacterial [1, 4, 6], UV protection [2], Waterproof with breathable properties [5], Electrical Conductivity [8], Thermal Conductivity [7], and so on [3].
Graphene has recently gained intense interest owing to its outstanding mechanical, thermal, optical, and electronic properties. Graphitic oxide is a raw material from which graphene is exfoliated (fig 1). Graphene is the two-dimensional sheet constituted by carbon atoms and creating a hexagonal lattice ordered. Graphene oxide (GO) is a graphene sheet with carboxylic groups at its edges and phenol hydroxyl and epoxide groups on its basal plane. Eliminating functional groups in the graphene oxide to produce reduced graphene oxide (rGO) by chemical treatment or by thermal annealing.

Fig 1: Graphene Oxide (Courtesy Jianchang Li et.al 2014)Waterproof breathable fabrics are engineered with the aim of protecting the wearer from weather conditions like wind, snow, as well as preventing excessive loss of body heat [9]. Waterproof breathable fabrics are one of the harsh weather fabrics that protect the wearer without hampering their efficiency. Garments made from such fabrics keep water from entering and wetting the body but allow the passage of air and moisture. The passage of water vapor from the garment makes it breathable and hence comfortable and it is one of the most essential attributes
that a garment should possess [10].
Various techniques can be used to produce waterproof breathable fabrics. One of the known methods of achieving waterproof-breathable fabrics is a pad-dry-cure method on the textile substrate. The incorporation of hydrophobic agents along with hydrophilic agents helps in the balance of the components and leads to better waterproof breathable properties along with other performance. This study will provide preparation and an assessment of waterproof breathable finish cotton fabric by using rGO and PUA.
Experimental procedure
Materials and methods
An average thickness (z) 1 – 4nm, lateral dimensional (X&Y) 5-10 µm, 1-3 number of layers, 220 m2/g surface area, >99% purity reduce graphene oxide (rGO) was purchased from Ad-Nano Technologies Private Limited, India. Polyurethane binder was supplied by Balaji chemicals, India. Scoured and bleached cotton woven fabric (200 GSM) supplied from Swadeshi Bleaching and Dyeing Mills Pvt. Ltd, India, and distilled water were used in the preparation of all solutions.
Preparation of breathable waterproof fabric
30 ml of distilled water was added with 0.5 gm rGO powder and kept in an ultrasonicator for 60 min at room temperature for getting dispersed aqueous rGO solution. rGO dispersed solution added dropwise into PUA binder under magnetic stirrer (500 rpm) at room temperature.
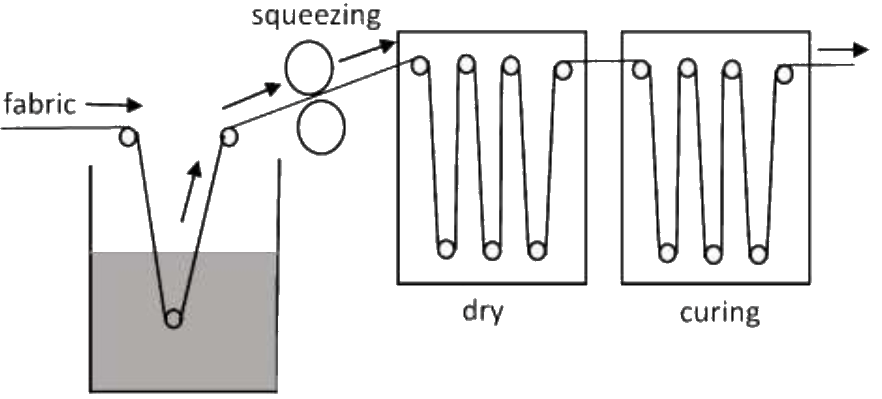
The fabric was padded with rGO/PUA mixture at 3 Kg/cm2 pressure then dried at 110oC for 3 min followed by curing at 150oC for 5 min in a lab center. Concentrations of rGO and PUA was mentioned below in table 1.
| Sample details | rGO% | PUA% |
|---|---|---|
| untreated | – | – |
| Sample 1 | 50 | 50 |
| Sample 2 | 60 | 40 |
| Sample 3 | 80 | 20 |
| Table 1: Concentrations of rGO/PUA mixture | ||
Apparatus & Characterization technique
The surface morphology and compositional analysis of the rGO thin-film material were carried out using a scanning electron microscope (SEM; 6510LA) with energy-dispersive X-ray spectroscopy (JOEL EDS System) facility attached to it, Fourier- transform infrared (FTIR) with wavenumber range of 500-4000cm-1, X-ray diffraction (XRD) with a monochromatized AI KR X-ray source at a constant dwell time of 100ms and pass energy of 40eV. Tensile strength was analyzed by using MTS Tensile Tester (Software – MTS Test works, the distance between jaws – 6 in, jaw speed – 12 inch/min, width – 1 in).
A Taber Stiffness Tester was used to perform the test with ASTM standard method D 5342-97 (ASTM method D5342). An electronic thickness tester, “Elektrophysik – MiniTest 600B” with a standard 526 µm ± 1% plate, was used to find out the thickness of the coated fabric.
Results and Discussions
Morphology of rGO material
The morphological characterization of the rGO material was illustrated in fig 3. Fig. 3a and 3c illustrate which layer by layer Graphene edges are observed and the flat sheet has lateral dimensions in the order of 5-10µm. It also is shown in fig. 3d fully exfoliated graphene material, some crumple on rGO is detected due to its atomic thickness in the range 1-4 nm.
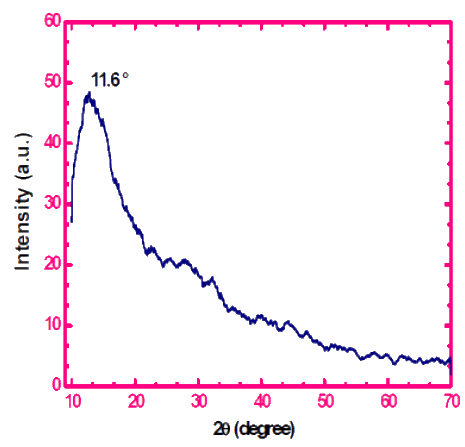
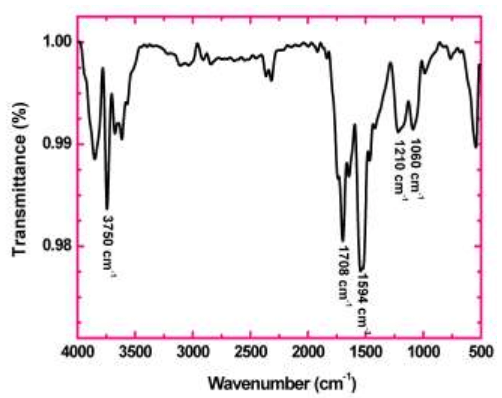
Furthermore, the XRD was analyzed to identify the crystalline region of rGO, in which at 2 was between 10-15 and the potter acute angle 2 was 11.40 of the reduced graphene oxide shown in fig 4. The peak of the graphite was observed as 26.40[11]. In addition, the FTIR of the rGO was also presented fig 5, which consisted of five different chemically shifted components that could be de-convoluted into OH groups (3400 cm-1), C=O (1740 cm-1), OH deformation peak (1420 cm-1), C-OH (1220 cm-1), C-O (1050 cm-1) and 1620 cm-1 assigned to the vibration of absorbing water molecules.

Waterproof with breathable property
Various tests were performed to determine the optimum combination for waterproof-breathable coating. Each result was analyzed and discussed in detail.
Spray Test
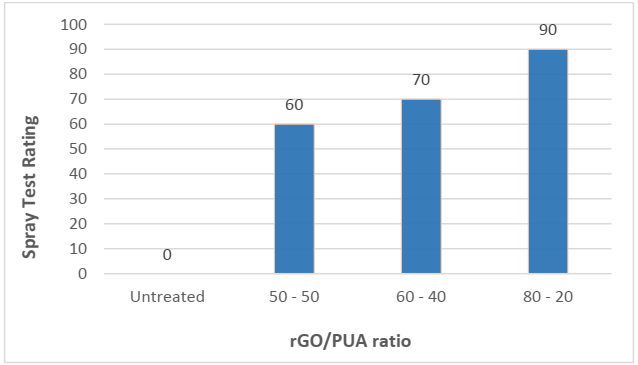
The spray test determines the water-resistance of the treated and untreated fabrics by using AATCC 22 (1996) test method. Fig 6 showed standard spray test ratings.

Spray test ratings are displayed in Fig. 7. In 80 – 20, ratio treated samples was achieved a better water resistance rating of about 90, as compared to 50 -50 and 60 – 40 ratio treated samples. In an 80 – 20 ratio coated sample, water droplets can be seen on the fabric surface which indicated waterproof property (Fig 8a) and the untreated fabric sample was completely wet on both surfaces (Fig 8b).
According to the spray rating chart, the 80-20 ratio of the rGO/PUA sample achieved the highest rating of 90 and this rating implies that there was “slight random sticking or wetting of upper surface”. It was observed that amongst these three samples, the sample from 80% rGO and 20% PUA showed maximum resistance to water and very slight wetting of the surface. It can be noted that as the amount of PUA was lowered, the water resistance was increased.


Contact Angle Test
Contact angle measurement helps in deriving the wettability of the surface. The higher the angle between the surface and water droplet, the higher the water resistance and the lower the wettability.
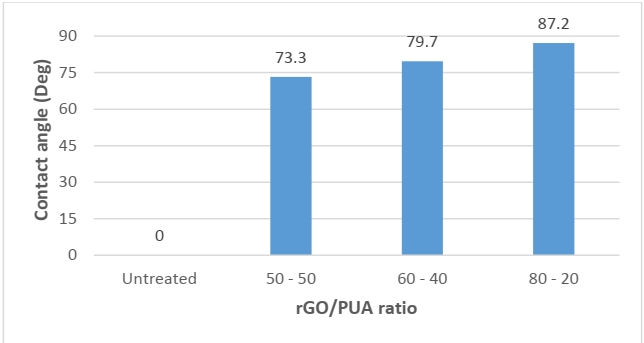
It was observed that the sample 80-20 ratio showed the highest contact angle 87.2o with the water droplet. Lower contact angle values were found in the case of 50-50 ratio which was 73.3o. In the case of uncoated fabric, the drop of water would seep into the fabric in 2-3 seconds.
Tensile Strength
The tensile strength test is performed to evaluate the mechanical properties of the fabric. During the coating process, the fabric might undergo changes in its physical properties due to tension and stretching. Sometimes the tensile strength of the fabric might get reduced due to the coating process. Hence this test ensures that no undesirable change has occurred in the strength of the fabric due to the coating process. The tensile strength was performed in both directions: warp and weft.
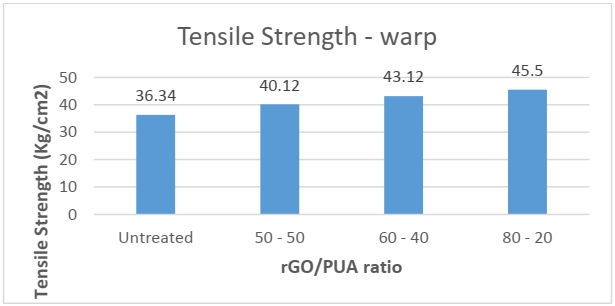

From the above results, it was observed that the tensile strength increased in both directions after the coating was applied. The maximum increase in average tensile strength in warp and weft direction was seen in the sample with 80-20 rGO/PUV composition. As the coat was applied using PUA which also worked as a binder imparted in the tensile strength of the fabric.
While reducing the concentration of PUA the strength increased this may be because of the rGO particles which are known to have higher mechanical strength. As the concentration of rGO increased it improved the strength of the coat indirectly resulting in higher tensile strength.
The rGO particles may have acted as particle reinforcement in the PUA whilst directly improving the tensile characteristics of the PUA, which can be seen in the results obtained.
Thickness Test
The thickness test was performed to evaluate whether an application of coating added undesirable thickness to the fabric.
| Sample No. | Composition (GO- PUA%) | Average sample thickness (µm) | Average coating thickness (µm) | Standard Deviation |
|---|---|---|---|---|
| Untreated | – | 287 | – | – |
| rGO/PUA-1 | 50-50 | 313.5 | 26.1 | 13.39 |
| rGO/PUA-2 | 60-40 | 325.7 | 35.7 | 15.35 |
| rGO/PUA-3 | 80-20 | 330.5 | 40.3 | 18.53 |
| Table 2: Thickness readings of chemical-coated samples | ||||
In the case of untreated fabric, the average thickness was calculated as 287µm. The sample composed of 50% GO and 50% PUA has the lowest average thickness, equaling 313.5µm. Furthermore, the highest thickness was found to be 3303.5µm in rGO/PUA which consists of 80% GO and 20% PUA. Lower values of standard deviation can be interpreted as less difference between the readings, which in this case means more evenness in the coating. In the case of Sample 1, which has the lowest average thickness, 313.5µm thickness was added to the fabric due to coating.
This is because it contains the highest amount of PUA, which is more fluid than the Graphene oxide resin. It was observed during preparation that the solution was difficult to emulsify due to the high amount of PUA. The solution was unstable and had high fluidity. During coating, the solution seeped through the fabric rather than being deposited on the surface. Due to this, there was a loss of the solution and hence the thickness was lower. The maximum amount of thickness addition can be seen in the sample with 80-20% GO/PUA, having 330.5 µm average
thickness. However, the standard deviation, in that case, is very high, which implies uneven coating.
Conclusion
From the above results, it is evident that the production of cotton fabric with waterproof with the breathable property as possible through rGO while PUV act as nucleation between the cotton fabric and rGO. The optimum combinations that yielded the best performance in terms of waterproof properties and breathability are samples with 80-20% rGO-PUA. These combinations can be used to create activewear that can be worn in harsh conditions as well as for regular wear apparel. Based on the standard test result of waterproof testing shows that increasing the concentration of rGO gives a better result with comfort property, as a result the concentration of making good breathable waterproof fabric was 80% rGO and 20% PUA.
Karthikeyan Saminathan, S.P. Sivasubramanian*.
DKTES’s Textile and Engineering Institute, Ichalkaranji
* Corresponding author: sp.sivasu@gmail.com
References
1. Andrea Zille, Luis Almeida, Teresa Amorim, Noemia Carneiro1, Maria Fatima, “Application of nanotechnology in antimicrobial finishing of biomedical textiles”, Materials research express, August 2015.
2. L. Qu, M. Tian, X. Hu, Y. Wang, S. Zhu, X. Guo, “Functionalization of cotton fabric at low graphene nanoplate content for ultrastrong ultraviolet blocking”, Carbon, (2014), 80, 565-574.
3. J.N. Chakraborty, Manas Ranjan Mohapatra, Jatin Kumar, “Differential functional finishes for textiles using graphene oxide”, Research Journal of Textile and Apparel, feb 2018.
4. Sundaresan K, Sivakumar A, Vigneswaran C and Ramachandran T 2011 “Influence of nano titanium dioxide finish, prepared by sol-gel technique, on the ultraviolet protection, antimicrobial, and self-cleaning characteristics of cotton fabrics”, J. Ind. Text. 41 259–77.
5. M. M., & Walsh, W. K., “AATCC Standards (2010) Method 22, Water repellency: Spray Test”, Research Triangle Park NC: American Association of Textile Chemists and Colorists Adler, 6. L. Karimi, M.E. Yazdanshenas, R. Khajavi, A. Rashidi, M. Mirjalili, “Using GO/TiO2 nanocomposite as a new route for preparation of electro-nductive, self-cleaning,
antibacterial and antifungal cotton fabric without toxicity”, Cellulose 21 (5) (2014) 3813-3827
7. Balandin, A. A., “Superior thermal conductivity of single-layer graphene”, Nano Lett. 8, 902–907 (2008).
8. M. Shateri-Khalilabad, M.E. Yazdanshenas, “Fabricating electro conductive cotton textiles using graphene”, Carbohydras, Polymer, 96 (1) (2013) 190-195.
9. Holmes, D. A., “Waterproof Breathable Fabrics”, In A. R. Horrocks & S.C. Anand (Eds.), Handbook of Technical Textiles, (pp. 282-315).
10. Krishnan S, “Technology of Breathable Coatings”, Journal of Industrial Textiles (1991), 21, 71-73.
11. Multifunctional cotton fabrics with graphene/polyurethane coating with far-infrared emission, electrical conductivity, and ultraviolet- blocking properties Xili Hu, Mingwei Tian, Lijun Qu, Shifeng Zhu, Guangting Han. Elesvier . August 2015.


Dear Sirs,
Could you advise we where I can source breathable water proof and fire resistant or laser safety fabric?