Color is an extremely important aspect of modern textiles. The color of a textile product is a major factor in the marketing and use of that product. The color of textiles can be used to differentiate groups of people such as uniforms used for athletic teams, hospital personnel, or military organizations. Color can also be functional such as camouflage or protective uniforms. However, in modern retail stores, the color of textile products is a major contributor to what is referred to as fashion. The color is very important with apparel, carpet, upholstery, curtains, drapes, sheets, and towels. All of the items are marketed with an emphasis on their specific color.
Textile Dyeing
Dyeing operations are used at various stages of production to add color and intricacy to textiles and increase product value. Most dyeing is performed either by the finishing division of vertically integrated textile companies or by specialty dyehouses. Specialty dyehouses operate either on a commission basis or purchase greige goods and finish them before selling them to apparel and other product manufacturers.
Textiles are dyed using a wide range of dyestuffs, techniques, and equipment. Dyes used by the textile industry are largely synthetic, typically derived from coal tar and petroleum-based intermediates. Dyes are sold as powders, granules, pastes, and liquid dispersions, with concentrations of active ingredients ranging typically from 20 to 80 percent.
Methods of Textile Dyeing
Dyeing can be performed using continuous or batch processes. In batch dyeing, a certain amount of textile substrate, usually 100 to 1,000 kilograms, is loaded into a dyeing machine and brought to equilibrium, or near equilibrium, with a solution containing the dye. Because the dyes have an affinity for the fibers, the dye molecules leave the dye solution and enter the fibers over a period of minutes to hours, depending on the type of dye and fabric used.
Auxiliary chemicals and controlled dyebath conditions (mainly temperature) accelerate and optimize the action. The dye is fixed in the fiber using heat and/or chemicals, and the tinted textile substrate is washed to remove unfixed dyes and chemicals. Common methods of a batch, or exhaust, dyeing include beam, beck, jet, and jig processing. Pad dyeing can be performed by either batch or continuous processes.
In continuous dyeing processes, textiles are fed continuously into a dye range at speeds usually between 50 and 250 meters per minute. Continuous dyeing accounts for about 60 percent of the total yardage of products dyed in the industry (Snowden-Swan, 1995).
To be economical, this may require the dyer to process 10,000 meters of textiles or more per color, although specialty ranges are now being designed to run as little as 2,000 meters economically. Continuous dyeing processes typically consist of dye application, dye fixation with chemicals or heat, and washing.
Dye fixation is a measure of the amount of the percentage of dye in a bath that will fix the fibers of the textile material. Dye fixation on the fiber occurs much more rapidly in continuous dying than in batch dyeing.
Each dyeing process requires different amounts of dye per unit of fabric to be dyed. This is significant since color and salts in wastewater from spent dyes are often a pollution concern for textile facilities. In addition, less dye used results in energy conservation and chemical savings. The amounts of dye used to depend on the dye are exhausted from the dyebaths which determine the required dyebath ratio.
The dyebath ratio is the ratio of the units of dye required per unit of fabric and typically ranges from 5 to 50 depending on the type of dye, dying system, and affinity of the dyes for the fibers. Dyeing processes may take place at any of several stages of the manufacturing process (fibers, yarn, piece-dyeing). Stock dyeing is used to dye fibers. Top dyeing is used to dye combed wool sliver. Yarn dyeing and piece dyeing, are done after the yarn has been constructed into the fabric.
Textile Coloration
Dyeing is the application of color to a textile material with some degree of fastness or permanence. The materials which impart the color are known as colorants. When these colorants have a natural affinity and permanence on textiles, they are referred to as dyes. Dyes actually migrate or diffuse into the chemical molecular structure of textile fibers in order to develop the final color of the textile product.
The dye-fiber molecular association is also responsible for the degree of fastness or permanence of the color because of the molecular attraction between the specific dye and the specific textile fiber; dyes are classified as being fiber specific. That is, dyes that work on cotton will not work on polyester, nylon, acrylic, wool, and many other commonly used textile fibers.
However, because the basic structure of cotton is cellulose, the dyes which work on cotton will also work on other cellulose-based fibers such as linen, ramie, rayon, and lyocell. It should be noted that the color actually produced on cotton by any single dye or combination of dyes will not necessarily match the color produced when these same dyes are used on linen or ramie or rayon. Many unique factors contribute to the color produced by textile dyeing including fiber properties. This situation will be discussed in more detail later in this document.
The other colorant used on textile fibers is pigmented. Pigments unlike dyes have no natural affinity for textile fibers. In order to produce permanent color on textile products, pigments are bound to the surface of the textile fibers using adhesives or glues which are known as binders. There is a wide variety of binder systems available with various properties. Their specific advantages and limitations will be discussed later. However, it is important to note that pigment colorants have no permanence on textile fibers without using some type of binder system. Additionally, many of the binders work on many different types of fibers. Unlike dyes, pigments are not fiber-specific. The same pigment binder combination can work on both cotton and polyester at the same time.
Historically, textile dyeing predates written history. There is evidence that indigo and other plant-based dyes were known and in use around 4000 BC. Modern countries such as Egypt, India, and China have archeological evidence of highly developed ancient textile products and processes. Mixing existing dyes to produce multiple shades in a wide color gamut was a common practice over 2000 years ago.
Natural Dyes
Until 1856, all textiles used throughout the world were natural dyes. That is, these dyes were obtained directly from the natural environment in one way or another. The vast majority of the natural dyes came from either plants or animals. For example, a major dye discovered by the Aztec or Mayan Indians was ‘cochineal’. It is a relatively bright red that is obtained from the body of the cochineal insect. The insect must be crushed and the dye is refined from the remains of the insect. Reportedly, 70,000 insects are required to produce one pound of dye. Cochineal is still used today as a natural commercial product in both textile dyeing and in food coloring.
Another natural animal dye that had a tremendous impact in the ancient world was ‘Tyrian Purple’. This dye was discovered near the ancient city of Tyre, which is in Lebanon. It was discovered that when a sea mollusk native to the Tyre coastal region was crushed and refined, an extremely bright purple dye was produced.
This dye exhibited very good fastness properties compared to other natural dyes. However, this dye was also very rare and difficult to produce. It took approximately 12,000 sea mollusks to produce one gram of dye. It should be noted that there are approximately 454 grams in one pound. Because of the brightness and rarity, Tyrian Purple was highly prized but also extremely expensive. Only the wealthiest in society, such as kings and queens, could afford this dye. From ancient times until today, the monarchies surrounding the Mediterranean Sea have purple robes as part of the royal heritage. Today, the Tyrian Purple sea mollusk is extinct, but the dye can be made synthetically.
The most well-known and most used dye of all time is ‘indigo’. This dye is obtained from the leaves, stems, and beans of a variety of plants, including one variety known as the indigo plant. In order to obtain the dye, the harvested plant matter is fermented over several weeks time. The dye itself is the major product of the fermentation process.
Figure 1 shows the indigo plants and beans, as well as, the fermentation process. After fermentation, the water-insoluble solid indigo is washed to remove impurities and dried. This results in a powdery solid dye. In order to dye textile fibers, especially cotton, the indigo must be made water-soluble by a chemical process known as reduction. The water-soluble reduced indigo is rapidly absorbed by the cotton fibers.
Once inside the fiber structure, oxygen in the air converts the indigo back to its original, water-insoluble form, as is illustrated above.


Modern indigo dyeing will be discussed in more detail later in this document. Although indigo was known by many ancient groups, the art of indigo dyeing of textiles was developed by the ancient Chinese and Indian cultures. Indigo-dyed cotton fabric was one of the major items of trade that prompted Columbus and others to search for new and better trade routes to India in the 15th and 16th centuries. Today, the modern textile industry uses synthetically produced indigo due to the high volume of dye required for current denim production. The natural indigo dye is too cumbersome to produce and requires enormous amounts of farmland. However, from the dyeing standpoint, synthetic indigo and naturally grown indigo are identical.
Synthetic Dyes
In 1856, Sir William Henry Perkin of England discovered the first synthetic dye, a purple known as ’mauveine’. His discovery not only revolutionized the textile dyeing industry, but it led to the development of the synthetic organic chemical industry. Indirectly, Perkin is responsible for modern medicines, plastics, paints, synthetic textile fibers, and many other modern products. Compared to modern synthetic dyes, natural dyes have poor color intensity and limited color durability. Also, these dyes can be very difficult to obtain, as well as cumbersome to apply to textile fibers.
Additionally, many of the natural dyes require metallic salts known as ‘mordants’ in order to produce some minimally acceptable degree of colorfastness. Many of these mordants are very toxic. Because of this fact, and other reasons, the use of natural dyes can actually be more damaging to the environment than the use of synthetic dyes. In terms of color performance and dyeing process control, synthetic dyes are fiber selective which, in part, accounts for their superior properties. However, the overall color quality of the final product can be influenced by a number of factors including fiber type and quality, dye selection and quality, purity of process water, and dyeing process control. These as well as other factors will be discussed in this document.
Textile Dyeing Preparation Processes
The quality of dyeing of any textile material is directly dependent on the quality of the textile fibers used to manufacture the product. Moreover, to maximize dyeing process efficiency and color performance properties, the textile fibers involved must be as clean as possible.
The central goal of preparation is the removal of both naturally occurring impurities and those which are added during yarn or fabric manufacturing. The dyeing process can be performed on textile fibers, yarns, fabrics, or garments, depending on the properties or for cost control. Preparation can also be performed at any processing stage depending on the dyeing technique employed. However, fabric preparation is typically most often used. Additionally, some commonly used optional preparation processes such as singeing or mercerization can only be performed on yarns or fabrics.
Complete fiber cleanliness is not easily achieved. The degree of cleanliness of the textile material is only determined by employing standard accepted testing methods and properly interpreting the data obtained. However, not all shades require the same degree of cleanliness to achieve high-quality color performance. Deep, dark shades of green, brown, blue, and black do not require the ultimate fiber cleanliness needed by whites, light pastel, and bright medium colors. Regardless, the often-quoted cliché’ still applies: “Well prepared is half dyed.”
Importance of Water Quality in Dyeing in Process
The purity of the processing water has a direct effect on the performance of the preparation processes which clean the fibers, the efficiency of the dyeing process, and the end product color properties.
Typical preparation processes used for cotton and cotton blend yarns, and fabrics include singeing, desizing, scouring, bleaching, mercerizing (cotton only), heat setting (synthetic fibers only), and cellulose enzyme treatments. Each process will be covered in this document. All of these except singeing and heat settings are processed using water baths.
Poor quality processing water creates all types of problems during the preparation and dyeing processes including color defects in the end product. Regardless of the source, the process water should be routinely analyzed. Key issues include water hardness, alkalinity or acidity (also known as pH), turbidity (suspended solids), sediment, dissolved organic matter, dissolved minerals such as iron, copper, and zinc, chlorine content, and color of the process water.
Water hardness is especially important for preparation cleaning processes. Hard water means that there is a large amount of calcium and magnesium dissolved in the water. The dissolved ions from the minerals can interfere with the performance of detergents, soaps, and wetting agents used in preparation. Hard water is typically responsible for the formation of soap scum or the ‘ring around the tub’ effect. This can sharply reduce detergent cleaning action. Additionally, iron or copper present in the water can have a very negative effect on bleaching. Specific effects of water-based impurities will be included in the discussion of the preparation processes.
As a general rule, it is important to keep in mind that just because the process water is clear, does not mean that this water is clean enough for preparation or dyeing. Also, if the process water is obtained directly from lakes, rivers, streams or wells, the quality of the water can vary from season to season throughout the year. Regionally specific water sources may require unique additional testing to ensure the acceptable quality of the process water.
As a general rule preparation processes, as discussed earlier, can be thought of as cleaning the textile substrate prior to dyeing, printing, and/or finishing. The essential objective of this process is to produce a substrate that has the following properties:
- even and rapid absorption of water,
- total removal of cotton seed husks, and
- the ability to absorb dyes and chemicals
Also, many end products are required to have high whiteness in order to be printed, dyed to a light or pastel shade, or to be sold as a market white. Preparation processes must be carefully controlled to minimize fiber damage which can lead to fabric yellowing or even strength loss.
Poor results from preparation processes can be due to poor process control and/or poor quality of the textile substrate. Often preparation problems are hidden or latent. That is, these defects do not appear in the substrate until after further processing. For instance, streaks or blotches seen in the fabric after dyeing may actually be the result of poor preparation. Also, fabric yellowing which is seen in storage may be due to fiber damage that occurred during the preparation process sequence. Preparation processes are key to good quality but are often overlooked.
Singeing or ‘Gassing’
Singeing is one of the few preparation processes which is not a wet process. In this process, a textile yarn or fabric is passed rapidly through a gas flame. This operation burns the fuzzy fibers from the surface of the yarn or fabric. The process is also known as ‘gassing’ the yarn or fabric.
The purpose of this process is generally to process a smooth yarn of fabric surface to increase the luster or the smooth feel of the final end product. With other products, the main purpose of this process is to reduce the pilling tendency of the end product. Pilling occurs when an end product forms small fiber balls on the fabric surface yielding a rough feel and an uneven appearance. Pilling is considered a quality defect. When surface fuzz is removed from the yarn or fabric, pilling tendency is typically minimized.
Figure 2 shows the results of the singeing process. Notice the results of the singeing process. Notice also that the yarns shown are plied yarns but singles yarns can also be gassed. Yarns are usually singed individually on a single end singer, then repackaged onto cones followed by further processing. These yarns typically are used for high-quality products.
Fabric singeing is more traditional than yarn singeing. In Figure 3, the fabric is dry, open width, and passed through the gas flames of the singer burners. These burners are essentially horizontal pipes with holes across the width of the pipe. Natural or propane gas is passed through the burner and ignited.


Figure 3 shows the schematic of a standard six-burner fabric singer. This burner configuration has three burners on the fabric front or face and three on the back. However, this burner configuration is variable. Most machines are capable of various configurations such as five burners on the face, one on the back, four on the back, with two on the face, etc. Key process variables include the speed of the fabric and the intensity of the gas flames.
Typically a sheeting weight fabric might be processed at speeds of 300-350 yards per minute (ypm) while heavyweight denim might run at 60-70 ypm. When yarns are processed, speeds can reach 1,000 ypm or more. However, yarn singeing is not as productive as fabric singeing and is considered a more costly and specialized process. The speed of the yarns or fabrics is a key processing control factor. If the fabric runs too fast insufficient singeing takes place. If the fabric runs too slow, it can be scorched or even burned. This is also true of singeing yarns.
Control of the flame intensity is an additional key control point. Also, when singeing blends of cotton and thermoplastic synthetic fibers melt balls can form on the tips of the synthetic fibers. These melt balls can cause the fabric to feel rough or raspy. They also dye deeper than the base fabric which can lead to a ‘specky’ dyeing appearance on light or pastel shades. So these fabrics are typically singed after dyeing. This is not an issue for 100% cotton fabric because cotton burns to fluffy light ash. When singeing after dyeing, care must be taken to select dyes that are not affected by the heat of singeing for these shades.
Once signed, there are usually glowing sparks or embers left on the yarns or fabrics. These must be cooled below ignition temperature to prevent further burning of the substrate. Usually, these sparks are quenched by exposing them to cold air, cold water-filled steel cylinders, or a water bath, such as a resizing or scouring bath which is used in a continuous fabric preparation range. Vacuum systems can be employed to remove burnt fiber ash from the singed fabric. However, washing the ash away during the subsequent preparation processes is the standard method to remove the ash residue.
Desizing
Desizing is a preparation process that is used only for woven fabrics. It is the wet process that degrades and removes the sizing materials which are applied to the warp yarns of a woven fabric. These sizing materials must be removed to enable the fabric to uniformly absorb dyes or finishing chemicals. Additionally, these materials, if not removed, can also interfere with various mechanical finishing processes resulting in poor end-product quality and/or finishing defects. This process is never used on knitted fabrics because sizing materials are not applied to yarns used for knitting.
The content of the warp size formulation including the chemical nature of the sizing materials is key factor in determining the type of desizing process used to completely remove these materials from the fabric. A typical sizing formulation can include a film former, such as vegetable starch, lubricants, binders, anti-stats, and waxes.
The film-forming component is normally the most difficult to remove. Vegetable starches like cornstarch or rice starch are mixed with water and cooked. They form a strong, smooth water-insoluble film. In order to desize these materials, they must be degraded to water-soluble components by using alpha-amylase enzymes. This is followed by rinsing the fabric multiple times with water to remove residue.
Synthetic sizing materials such as PVA (polyvinyl alcohol), CMC (carboxymethyl cellulose), and PAA (polyacrylic acid) are normally water-soluble. These materials can usually be easily removed by using a warm or hot washing bath which includes detergent and alkali. The specifics of the time and temperature of processing depending on the exact nature of the synthetic material and the amount of it that is present. Specifically, PVA is often captured from the desizing process and recycled. The vegetable starches cannot be recycled while the other synthetic sizing materials are not cost-effective when recycled.
Desizing can be accomplished in a batch dyeing machine using water at a temperature range of 40-60 minutes. This process can also be part of an open-width continuous preparation range. It often follows singeing. The desizing chemical bath is used to quench the sparks and embers from singeing.
The saturated fabric then travels into a fabric steamer where it is allowed to dwell for 10-20 minutes to allow the degradation of the sizing materials to occur. These components subsequently are removed by rinsing the fabric in a series of fabric washers. An important quality control factor is to test for residual sizing materials. There are various specific tests published for this process depending on the chemical nature of the sizing materials.
Many companies place a drop of iodine on the surface of the desized fabric. If the iodine turns blue, this is an indication of the presence of starch. Further testing or reprocessing of this fabric may be required. Other spot tests are used for the other sizing materials. The desized fabric may then be held in the wet or dry state waiting for further processing. However, the fabric may go immediately into the next preparation process which is scouring.
Scouring
Scouring is the process, used on both knits and wovens, where the impurities in the fabric are removed by washing. Usually, the only major impurity not removed by this process is the inherent color of the fiber.
Table 1 gives a list of the various impurities found in typical cotton substrates. The exact type and amount of these impurities can vary due to factors such as fiber quality, fiber harvesting methods, textile substrate storage, and handling as well as many other factors.
| Cellulose | 88.0 – 96.0% |
|---|---|
| Proteins | 1.1 – 1.9 % |
| Pectin Substances | 0.7 – 1.2% |
| Ash (Ca, Mg, K, Na, Phosphates, Carbonates) | 0.7 – 1.5% |
| Organic Acids | 0.6 – 1.0% |
| Wax | 0.4 – 1.0% |
| Sugars | 0.1 – 0.4% |
| Other (dirt) | 0.5 – 1.0% |
Table 1 – Components of Raw Cotton Fiber by Percentage.
These specific types of impurities and the approximate amounts are found in bales of cotton fibers. These impurities will pass through the yarn and fabric manufacturing processes. In addition, the textile substrate will also include many of the mill impurities listed in Table 2. All of these impurities should be thoroughly removed prior to dyeing or their presence can lead to poor dyeing quality, poor finishing performance, and end product defects. Unlike desizing, scouring is absolutely necessary for all fabrics and all dyed shades.
Impurities During Manufacturing Process
- Mill Impurities
- Fiber Finishes
- Tints
- Sizes
- Chalk Markers
- Lubricants (fiber and yarn)
- Oils
- Greases
- Dirt
- Rust
- Metals in Lubricants
The basic process involves using detergents and alkali to remove these impurities by washing. Like desizing, this process can be accomplished in batch form using standard dyeing equipment or as a section of a continuous preparation range. Many of the impurities listed in Tables 1 and 2 are not water-soluble.
For cotton, this is especially true of the outer waxy layer which, if not removed, will not allow water to penetrate the fiber. This produces serious negative effects on subsequent water-based processing such as dyeing or chemical finishing. Detergents have the ability to allow oily, greasy liquids and solids to mix with water. This dirty water is then drained from the dyeing equipment or washer and sent to the wastewater treatment system for final disposal.
Typical washing temperatures range from room temperature (70oF) to boiling (212oF). However, most processing is done at 120o-180oF. The fabric must be mechanically agitated during the process. In the case of continuous processing, the fabric is saturated with the scouring bath. The fabric then goes into a steamer where it dwells for 20-40 minutes. In both cases, the scoured fabric must be thoroughly rinsed multiple times to ensure the maximum removal of residual impurities.
It should be noted, most companies mix their own scouring formulations from basic chemicals to match the nature of their specific substrates. This is done to maximize cleaning efficiency while minimizing costs. Greige, unscoured substrates should never be stored laying on the floor of the plant or handled in a manner so that they pick up impurities. These unknown impurities add uncontrolled variables to the process and may not be removed during processing. This can lead to various product defects and overall poor end-product quality.
The results from a well-executed scouring process include:
- a uniformly clean base for dyeing yarns and fabrics in all shade ranges,
- removal of fats, waxes, oils, greases, and dirt,
- better textile substrate absorbency resulting in more uniform subsequent processing,
- softening and preconditioning cotton motes for removal in bleaching, and
- a clean, neutral base for finishing
Scouring is the single, most critical, a process in preparation. Insufficient scouring results in poor performance in many subsequent processes; especially dyeing and chemical finishing. Residual impurities left from the poor scouring result in dye blotches, streaks, and spots, as well as, insufficient finishing performance. Residual oils left on the fabric can degrade during storage resulting in fabric yellowing. Additionally, degrading impurities can also result in the formation of noxious fabric odors. This can lead to poor end-product performance and strained customer relations.
The effectiveness of a scouring process is normally evaluated by using a laboratory method that involves the extraction of residual oils, greases and dirt. The amount of residual materials extracted is correlated to the degree of cleanliness of the substrate. However, companies often use a quick method where a few drops of clean water are placed on the dry, scoured fabric surface. The time taken for the drops to be absorbed is recorded. Normally, clean scoured fabrics should absorb the water in 1-3 seconds.
Bleaching
For pastel or bright shades, including market whites, scouring alone does not yield a sufficiently clean textile substrate. These products require bleaching, which is the preparation process where the inherent color of the fabric is removed. It is a destructive process where the color-producing molecules are destroyed by oxidation. This process also destroys oil and dirt residues left from the scouring process.
After properly bleached, 100% cotton substrates are 99.9% plus pure cellulose and are the cleanest substrates produced by preparation. However, the textile fiber can also be attacked by the bleaching process resulting in fiber damage. This fiber damage can be severe if the process is not well controlled. Bleaching is a balancing act between fiber cleanliness and whiteness versus potential fiber damage.
The oxidizing agents used for this process are known as textile bleaching agents. In the modern textile industry, there are two main bleaching systems employed. The most widely used bleaching agent is hydrogen peroxide. This agent uses a form of oxygen as the oxidizing agent in the process. The other popular bleach is sodium hypochlorite and uses chlorine as the bleaching agent. The main goals of the bleaching process are to obtain a white, ultimately cleaned substrate in the shortest time (highest productivity) possible with minimum fiber damage. The goal is the same regardless of the bleaching agent chosen.
There are distinct differences between hydrogen peroxide and sodium hypochlorite. Hydrogen peroxide is the more expensive chemical on a cost per pound basis. However, it is the most popular bleach due to its versatility. Hydrogen peroxide can be used with virtually any fiber as long as the process conditions are carefully controlled. It also exhibits wide process temperature versatility being effective at low temperatures such as 120oF and high temperatures such as 212oF (boiling water). The temperature actually used in the process will depend on the type of fiber blend, the concentration of the bleach in the formulation, and the degree of whiteness required by the end-use.
Hydrogen peroxide is also the most environmentally friendly bleach. It is a very strong oxidizing agent and must be chemically stabilized during the process in order to prevent poor bleaching results. However, after the process, hydrogen peroxide will chemically break down in the wastewater to form only oxygen and water. These components not only are not polluting, but they actually improve the performance of wastewater treatment systems. Under the correct bath conditions, hydrogen peroxide is also safely used on fiber and yarn-dyed products. It is widely known as the “color safe” bleach. However, this is not absolute and should be evaluated on a dye lot to dye lot basis.
Sodium hypochlorite is less expensive than hydrogen peroxide. It is an excellent bleaching agent for cotton and other cellulose-based fibers. However, it is not nearly as versatile as hydrogen peroxide. It is well known that this bleach causes fiber yellowing when used on nitrogen-containing fibers such as spandex and wool. It also causes fiber damage due to the retaining of chlorine by the nitrogen in the fibers. This bleach destroys or severely damages most dyes that are used on cotton or cotton blends. It is not a “color-safe” bleaching agent.
It also has much more temperature and pH sensitivity than does peroxide being most effective at temperatures between 90-140oF. After bleaching an additional agent known as an antichlor must be added to one of the rinsing baths to prevent unused bleach from having negative effects on subsequent processing. In bleaching wastewater, sodium hypochlorite is a pollutant that must be carefully neutralized. Chlorine in the wastewater treatment system can form organochloro compounds which may be toxic. Also, improper waste treatment can liberate chlorine gas which is toxic and dangerous.
Both peroxide and hypochlorite bleaches can cause severe fiber damage during the bleaching process if the textile substrate contains metal contamination such as iron rust or copper shavings. This fiber damage can be so severe that holes can occur in the substrate where the metal particles exist prior to bleaching. This problem is due to the fact that these metals act as a catalyst for the bleaching action, accelerating it to the point that it severely weakens or even destroys the fibers during the process.
The best way to prevent this problem is to handle the substrate carefully to avoid contamination from rust particles, metal railings, or rusty drip marks from plumbing pipes. Maintenance personnel should be trained to avoid metal shavings contamination on the substrates when processing equipment is serviced. Dissolved metal contamination in processing water is also a major problem. To counteract this problem, a sequestering agent, which keeps dissolved metal ions from interacting with the bleach, is added to the bleaching formulation. Other agents added to a typical bleaching formulation include penetrants, bleach stabilizers, alkali, and pH buffering chemicals. The buffering chemicals are used to keep the bath between pH 9-11 during bleaching which is optimum for most formulations.
As with scouring and desizing, bleaching can be accomplished with batch equipment or as a step in a continuous preparation range. Key control parameters for the bleaching process include time, temperature, and concentration of the bleaching chemicals. Generally, with batch bleaching, the higher the temperature the shorter the time or the higher the degree of bleaching. Also, the higher the bleach concentration the shorter the bleaching time and the potential for more severe bleaching action. Over-bleaching causes fiber damage which often results in fabric yellowing and loss of fabric strength.
With continuous bleaching, the fabric is saturated with the bleaching bath, followed by steaming. The length of time in the steamer will be determined by the fiber content, the concentration in the bleach bath, and the degree of whiteness required by the end-use. Typical steamer dwell times vary between 15-40 minutes for most cotton products. Once out of the steamer the fabric goes through multiple washing steps to remove residual bleach and impurities.
In the case of some pastel or bright shades or market whites, the white obtained from bleaching may not be sufficient. In those cases, a fluorescent whitening agent (FWA) or optical brightening (OBA) may be added to the bleaching bath or in a rinse bath after the bleaching process. The FWA’s or OBA’s are colorless organic compounds that absorb invisible ultraviolet radiation and emit this energy in the visible light region. This property is known as fluorescence.
Figure 4 illustrates the fluorescent nature of FWA’s or OBA’s. Thus, the amount of light energy reflected from a textile substrate can actually exceed 100% of the incoming light energy illuminating the material. These materials have the ability to make white textile materials appear “whiter than white.” In the same way, they can make dyed materials appear “brighter than bright.” However, OBA’s are quality improvement compounds used with bleaching but they are not a substitute for the bleaching process.
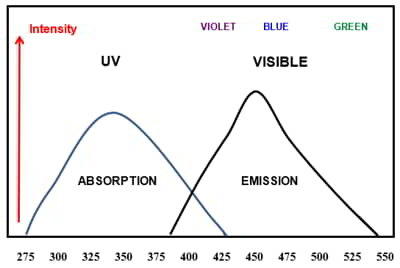
These FWA’s or OBA’s have varying chemical natures and are often fiber specific. Anionic OBA’s which have a negative electronic charge in water are used with cotton, wool, and nylon. Cationic OBA’s which have a positive electronic charge in water are used with acrylic and some polyester fibers. Nonionic which have no electronic charge can be used with most synthetic fibers. These compounds vary in their fastness to washing and daylight. They usually have poor wash-fastness but can be reapplied. Most commercial detergent formulations include a mixture of optical brightening agents and these are applied in the standard home laundry.
Mercerization
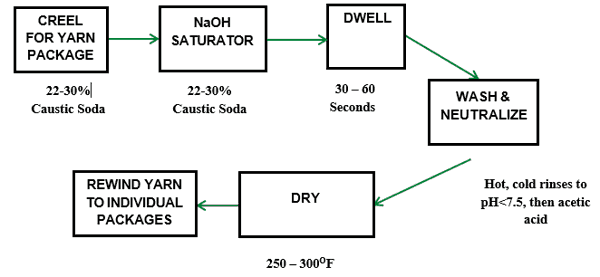
Mercerization is an optional quality improvement preparation process. It is used almost exclusively for cotton. It is one of the oldest modern finishing processes and was developed for cotton substrates by an English chemist, John Mercer in 1857. Today it is mainly used for cotton fabrics but can also be used for cotton yarns. This process involves the treatment of cotton yarns or fabric held under tension while being treated with a strong sodium hydroxide solution.
Figure 5 shows a schematic of the mercerization process. In the initial section of the range, the fabric is impregnated with sodium hydroxide solution. In this example, a 22-30% solution of caustic soda (sodium hydroxide) is applied to the cotton fiber held under tension at room temperature. The caustic soda causes the fiber to swell and the internal cellulose molecules rearrange to form the ‘Cellulose II’ structure from the standard ‘Cellulose I’ configuration. The subsequent swelling of the fiber molecules collapses the lumen. Then the fabric is passed over timing rollers to allow the caustic enough dwell time at ambient temperature to accomplish the process. This step is also called ‘skying’. The total dwell time of the caustic soda in the fiber prior to washing is normally 30 seconds to one minute.
In the stabilize section, the fabric is held by the edges in order to exert the tension necessary for the full development of the final mercerized fabric properties. Cascade rinsing is a counter-flow washing process where the cleanest water is introduced through the last cascade unit. The water in this process is heated to 200oF to remove the caustic soda. The first cascade washer uses the water which is most saturated with caustic. This “dirty” water is captured after passing through the fabric. The caustic soda is separated from this wastewater and recycled. The mercerized fabric is thoroughly washed and any residual alkali is neutralized. The fabric may be stored wet or dry prior to dyeing and further processing.
Once the caustic soda is removed by thorough washing, the cotton substrate is dried and returns to approximately its original size but in a different cross-sectional shape.

Figure 6 is an artist’s concept of the changes in the cross-sectional shape of cotton fiber during the mercerization process. Notice the non-uniform uneven appearance and various shapes of the unmercerized fibers. Then note the uniformity of the rounded shape of the mercerized fiber. Also, it can be seen that the lumen has collapsed to various degrees.
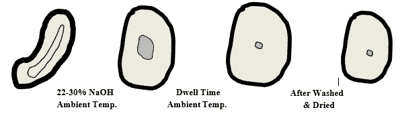
Figure 7 is an actual microscopic photo of cotton fibers before and after the mercerization process.
The positive results of the mercerization process include increased dyeability and uniformity. Dye yield by the mercerized fibers can be as much as a 20-25% increase in shade depth over unmercerized. This can be major cost savings because a given shade may be obtained using approximately 20% less dye.

Mercerized cotton substrates show up to a 20% increase in tensile strength. These products are normally dimensionally stable and absorb water and chemicals more rapidly than non-mercerized substrates. Mercerization significantly improves the luster of the product because the substrate is kept high tensions. The fiber surface becomes significantly smoother and reflects light in a mirror-like fashion.
Many bales of cotton have some immature fibers. These fibers have a primary wall but due to some growing conditions, they did not grow a significant amount of cellulose in the secondary wall. Some immature fibers have no secondary wall cellulose and will not dye and appear as white specks in dyed fabric. A name often applied to these fibers is ‘dead cotton’. Figure 8 shows immature cotton before treatment. Mercerization is the only process commonly used to remedy problems resulting from immature fibers.
This process will swell any small amount of cellulose present in the immature fibers so that they will be dyeable as is shown in Figure 9. Any non-cellulosic material in the fabric will also be destroyed and washed away during processing.


Yarn mercerization uses essentially the same type of mercerizing bath and similar processing conditions. However, yarn mercerization is a more complex process. The yarn is taken off creels to form yarn ropes which go through the mercerizing process. These ropes must then be dried and the mercerized yarns must be individually rewound onto packages prior to fabric manufacturing, typically knitting. Yarn mercerization is only possible with plied yarns due to the high tensions used to create luster.
Best results are obtained when the yarns are singed (gassed) prior to mercerization. These yarns can be dyed prior to knitting or dyed after knitting in fabric or garment form. These yarns are often mercerized in fabric form after knitting. This produces a cotton product with extremely high luster. This product is known as ‘double mercerized’. Figure 10 shows a schematic of the yarn mercerizing process.
Heat Setting
Thermoplastic synthetic fibers such as polyester or nylon are greatly affected by heating them above their glass transition temperature (Tg). This temperature is unique to each fiber type and variant. When the fiber is heated above its Tg but below its melting or degradation point, the internal fiber molecules flow and rearrange along lines of tension while the outside of the fiber remains solid. This process allows the manufacturer to set the dimensions of the substrate minimizing shrinkage. Special effects can be set into fabrics such as permanent designs or crinkles. In garments, permanent pleats and creases or a specific desired shape can be heat set into the textile product.
However, cotton and other cellulose-based fibers are not thermoplastic and not affected by the typical heat setting process. Exposure to high temperatures above 400oF for long periods of time may scorch cotton and result in product degradation and yellowing. In the special case of spandex blends, heating processes are used to stabilize the spandex fiber prior to wet processing. After wet processing spandex blends are processed through a final heat treatment to set the amount of stretch in the final product. Great care must be taken in processing spandex blends in order to produce uniform product properties with satisfactory performance.
The dyeing performance of polyester and nylon is directly affected by heat settings. Uneven heating across the width of the tenter frame oven leads to uneven heat setting across the width of the fabric. Therefore side-center-side dyeing (shading) defects are heat set into the fabric. The uneven moisture content of the fabric entering the oven also can lead to non-uniform processing. The typical Tg for polyester fibers is approximately 330-350oF. For nylon, Tg is around 390-410oF. The Tg needs only to be exceeded for a few seconds for a uniform heat setting to take place. Cotton/polyester and cotton/nylon blends are normally heat set to obtain maximum dyeing uniformity of the synthetic component and acceptable product shrinkage properties. Heat setting can also be performed as a mechanical finishing process after dyeing. Dyes must be selected and evaluated to withstand this high-temperature process if heat setting after dyeing is used.
Cellulase Enzyme Processing
Cellulase enzyme processing is an optional preparation and/or washing technique that removes surface fiber fuzz and softens the feel or hand of the fabric or garment. Enzymes are organic substances that quicken certain natural reactions. They are usually obtained from natural sources such as bi-products from a biological process like fermentation. These enzymes are usually easily biodegraded in wastewater and thus are considered environmentally friendly processing chemicals.
Cellulase enzymes have the ability to selectively hydrolyze or dissolve cellulose. They are used for surface fiber fuzz removal, color wash down of specific types of dyed fabric, and increased fabric softness. However, fabric weight reduction with subsequent fabric strength loss results when the enzyme action of the process is too severe or is allowed to continue for too much time.
Figure 11 illustrates the improved fabric appearance obtained from cellulose enzyme treatment. The enzyme process removes the surface fiber fuzz. The treated fabric appears smooth and clean. It also appears deeper in color and brighter in shade. Notice also that the surface of the treated fibers appears much smoother in the microscopic photo.


The overall enzyme action of the process will depend on many factors including the type of enzyme chosen, the temperature of the bath, the liquor ratio of the bath, the concentration of the enzyme, the type and nature of auxiliary chemicals used, the pH of the bath and the overall processing time. Enzymes are very sensitive to bath pH, temperature, and processing. They work within a specific temperature range. Outside of these upper and lower limits, very little enzyme action is produced. Additionally, some enzyme types are more aggressive than others.
Note that the acid cellulose reached peak activity at pH 4.3 while the neutral cellulose reached peak activity at pH 6.0. Both enzyme types exhibit reduced enzyme activity outside of their optimum pH range.
One other major factor in cellulase enzyme processing is the mechanical agitation of the processing equipment. Typically cellulase enzyme processing is accomplished in batch dyeing machines or garment washing machines such as the rotary drum machine. The mechanical agitation improves the removal of hydrolyzed material. There are limits but in general, the higher the mechanical agitation level, the better the removal of hydrolyzed residue. This results in reduced processing times and improved process performance.
There are a few key parameters for optimizing the overall preparation processing. The operation must be systematic and consistent. Manufacturers should thoroughly understand their fabric properties and the capabilities of their processing equipment. All equipment should be properly serviced and maintained. All processes should be routinely monitored and published procedures should be closely followed. Process operators should be thoroughly trained and properly supervised. Well-prepared textile substrates are absolutely necessary in order to produce high-quality end products.
Color and Shade Matching
The ability to perceive color is an ability controlled by human genetic make-up. Color vision is part of human vision but is not totally understood. Basically, it is believed that color vision is due to light energy, electromagnetic radiation, striking nerve endings located in the retina of the eye causing complex photochemical reactions to occur. This stimulus sends a signal through the optic nerve to the brain. This signal produces the information that the brain interprets as color vision. However, this is a very complex process. Color vision also seems to be affected by the emotional state of the person. People use the perception of color to describe emotional situations.
Most everyone has used terms such as ‘a gray day’, ‘blue Monday’, ‘black Friday’, and ‘I was green with envy. Therefore, the ability to perceive color can be described as a psychological- physical phenomenon.
Some people confuse certain colors. This condition is known as color blindness. The most common form of color blindness is when people confuse reds and greens. Although well known, very few people have the colorblind condition of confusing blues and yellows. Even fewer people are completely color blind, that is, they only perceive black, white, and various shades of gray. Proper color vision testing is the only way to accurately determine color perception capability. People who are not tested as color normal should not be involved with the shade matching of dyed textiles.
Color Perception and Light Energy
Based on the previous description of color vision, it is obvious that without light energy there is no perception of color. Described in a different way, if all light is removed from a room, all the objects in the room become the same. That is, if there is no light, there is no color.
Visible light is energy that causes stimulation of the nerve endings in the human eyes to produce the sensation of vision and color vision. Light energy is a narrow band of the broad spectrum of electromagnetic radiation produced by stars. This includes lethal radiations such as cosmic rays or x-rays. It also includes harmful radiation such as ultraviolet or microwave energy. Additionally, it includes harmless radiation such as radio waves. People have learned to produce certain radiation, such as microwaves or radio waves, and develop useful devices such as microwave ovens.
The current scientific description of electromagnetic radiation states that all of these energies move at the same speed, the speed of light. All of these energies move through space in the form of waves described as ‘sine’ waves. Radiations types are separated by their individual wavelength. Wavelength is the measured distance from the top of one sine wave to the top of the adjacent wave. Cosmic rays, which are lethal, have very short wavelengths while radio waves, which are harmless to humans, have very long wavelengths. For uniformity, the wavelengths are normally stated in units of meters. The range of wavelengths of visible light which is also harmless is from 380-780 X 10-9 meters. This unit is converted to a new unit called nanometers.
Figure 12 shows that different groups of visible light wavelengths produce different color sensations. When these groups are spread out so that these wavelength bands can be seen, we call this array the visible color spectrum. The combination of all of these wavelengths produces white light energy. Wavelengths shorter than 380 nanometers are not visible and fall into the ultraviolet radiation range. Wavelengths longer than 780 nanometers are also not visible and fall into the infrared radiation region. Colors produced by these individual wavebands of energy are known as spectral colors. Colors that are produced by mixtures of these wavelengths are known as non-spectral colors. The special case of black, white, or gray sensations is known as achromatic colors.
| ƛ (mu) | COLOR |
|---|---|
| 380 | UV |
| 380 – 435 | Violet |
| 435 – 480 | Blue |
| 500 – 560 | Green |
| 580 – 595 | Yellow |
| 585 – 650 | Orange |
| 650 – 780 | Red |
| 780+ | IR (near) |
Figure 12 – Hues of Monochromatic Light
Color Perception and Objects (Textile Substrates)
Textile substrates produce no color of their own, unlike sources of light energy. Textile substrates require an external light source or illuminant to produce a color sensation. This fact is illustrated in Figure 13.
From this schematic, when incoming light energy strikes a textile fabric (substrate) various results can occur. First, all of the light energy can be reflected, similar to a mirror. Nearly all textile fabrics reflect only a portion of the incoming light. Alternatively, all of the light energy could be absorbed by the fabric.
Textile fabrics only absorb a portion of the light. Only a black hole in space completely absorbs all light energy. In general, a portion of the light energy is reflected and the remainder of the light is absorbed. However, especially with sheer fabrics or substrates, there is the possibility of light energy being transmitted through the fabric. In this case, the fabric acts as a light filter.
Sheer fabrics are important textile products; however, in terms of color evaluation, the color of the fabric can be misread when transmitted light from the color of the background is viewed through the fabric. For color evaluation, sheer fabrics should be folded until no light can be transmitted through the fabric so that only reflection and absorption can occur.

Basically, it is the interaction of the light energy, striking the fabric, with the dyes that are in the fibers of the fabric or the pigments glued on the surface of the fabric that produce the color sensation. Incoming white light energy contains all the color wavelengths. When this white light strikes the fabric, the dyes absorb certain color wavelengths and reflect others. The color of the textile fabric is due to the combination of wavelengths reflected. When several dyes are mixed together, the color of the textile substrate will result from the combination of color wavelengths reflected.
The structural nature of the fiber and its cross-sectional shape, as well as, the geometric structure of the textile substrate have a direct influence on the final color of the product. For example, the color of trilobal nylon 66 fiber will appear to be somewhat different from the color of round nylon 66 fiber when they are dyed together. The color of 18/1’s ring-spun 100% cotton yarn will appear to be different from that of 36/2’s ring-spun 100% cotton yarn.
The color of the cut pile carpet will appear to change as the direction of the pile is changed. All of these color differences are due to the different angles of light reflection from the different surface geometries. Additionally, cotton and rayon are both pure cellulose-based fibers and will dye with the same dyes. However, when textile substrates composed of the two fibers are dyed together the color produced on each substrate will be similar in shade but not match. This color difference is due to the different internal molecular configurations of the two fibers. Similar results are seen with other natural fibers and also with synthetic fibers.
Color Mixing and Shade Matching
Today, color is produced by placing dyes and pigments on textile substrates. Color is also produced by colored light sources. In either case, single or primary colors are mixed to produce a wide variety of colors known as a color gamut or range. However, the specific mixing technologies are quite different between colored light and dyes. In the light mixing system, the primary colors are red, blue, and green light.
When red and blue light are mixed, magenta or violet is produced. When blue and green light is mixed, cyan or turquoise is seen. When red and green light is mixed, yellow is produced. The colors produced by mixing, magenta cyan, and yellow, are brighter than the primary colors because light energy has been added together. This is known as the ‘additive color’ mixing system. When all the primary colors are mixed together in the proper proportion, white light is produced. The sensation of black is produced by the total absence of light energy.
For the dye (pigment) mixing system, the primary colors are red (magenta), blue (cyan), and yellow. Magenta and cyan are the scientifically true primary colors; however, modern dyehouses use red and blue dyes which are used for most of their production shades as their color primaries. When red and blue dyes are mixed, purple is produced. When red and yellow dyes are mixed, orange is produced. When blue and yellow dyes are mixed, green is produced.
The mixture colors, purple, orange, and green are duller than the primary dyes because they subtract more light energy from an illuminating light source than the primary dyes. Therefore, this system is known as the ‘subtractive color’ system. Black is produced by mixing the proper portion of each primary dye. Interestingly, black is known as the ultimate recovery color for textile products. Virtually, any off-quality dyeing can be overdyed into a first quality black shade. However, the quality of the black shade is highly dependent on the skill of the dyeing company. There are no white dyes. White on textile substrates is produced by the absence of any dye.
On most textile fibers, white is produced by bleaching which destroys any color-producing material in the fiber. The resulting white sensation is due to the light-scattering properties of the textile fiber. However, white is produced by pigment whitening agents such as titanium oxide, TiO2. Pigment whites are often used in printing. TiO2 is extruded with certain synthetic polymers to produce white fibers. Otherwise, these fibers would be clear.
The purpose of dyeing is to produce color in textile substrates. However, the specific shades produced are not an accident of processing. Textile substrates are dyed with a specific dye formulation to match a color standard. Color standards are a part of the overall specifications of the textile end product. Color standards are generally agreed to by both supplier and customer. The details of color standards development and the specifics of shade matching are beyond the scope of this document. However, certain basic facts should be understood.
Visual shade matching is still the most widely used method for textile substrates. Professional shade matches should be routinely tested for color blindness and properly trained in shade matching techniques. They should understand the various properties of the dyes and textile substrates with which they are working. Shade matching should take place using standard light sources and employing a neutral gray background. The specific geometry of textile substrates along with any influence of surrounding colors on the color match should be known and taken into account. Visual shade matching is a complex operation that requires extensive knowledge and a high degree of skill.
In recent years, color measuring instruments or color computer systems have proven to be highly valuable. There are shade matching programs such as ‘Ecmc‘ which have produced reliable results. The typical way these systems work is to illuminate a colored substrate with a standard light source. The amount of reflected light energy from the substrate is measured wavelength by wavelength across the visible spectrum. This reflection data is used to calculate a color difference or ‘E’.
For most textile products E= 1.0 is the typical pass-fail boundary for shade matching. However, this boundary can be higher or lower depending on the fabric construction and fiber content of the end product. Color instruments only measure one color at a time and have difficulty with multicolor designs such as stripes or plaids. They also do not automatically account for substrate texture. Therefore, it must be kept in mind that color computers measure data and calculate color. People see color. These two are not the same. It should also be noted that the limits of an acceptable shade match are often business-dependent. When business is good, shade tolerances for many textile products are wider. When business is not good, these tolerances can be tighter.
Color computers, when programmed with the proper software, can use reflectance data to calculate predicted dye formulas. They can also be a useful tool to store specific information concerning dye properties, dye costs, mixing procedures, and dye combination issues. These instruments can monitor overall dyeing quality. Many companies use color computer systems because the systems allow colors to be matched at lower production costs. The use of these instruments also helps improve overall process productivity.
Dyes and Dyeing of Textile Substrates
Dyes are the chemicals that are absorbed into the molecular structure of textile fibers which produce the color of the molecular structure of textile fibers which produce the color of the textile product. Dyeing is the process that places the dyes inside the fibers. Currently, dyeing of textile substrates takes place using water baths. Dyeing equipment is used to contain the textile substrate and the dye bath during the dyeing process. The dyeing equipment is also used to control the necessary parameters of the dyeing process in order to maximize dyeing quality and productivity. Pigments are a special case of textile coloration. They require a binder or glue to adhere them to the surface of the textile fiber. Not all dyeing equipment can be used to successfully apply pigments to textile substrates.
General Dyeing Factors
The primary objective of the dyeing process is to produce the shade on the textile substrate that matches the color standard. If the substrate is off-shade, it is of quality by definition. Next in importance is to produce the shade with the colorfastness properties which meet the performance specifications required by the end-user. For example, most garments must be able to be washed without color bleeding or fading. Outdoor furniture upholstery fabric should be highly resistant to light fading as another example. Each dye formulation must be evaluated for its colorfastness properties. Finally, dyeing companies are for-profit businesses.
The cost of the dyeing process both in terms of dyes, equipment, and processing times, greatly impacts the profitability of a manufacturing company. Other key factors considered when selecting dyes include ease of mixing with other dyes and chemicals, uniformity or level dyeing properties, dusting issues when using powdered dyes, and overall environmental impact. Environmental concerns are of particular importance. In some cases, certain dye formulations produce large volumes of highly colored wastewater. In this case, the volume and intensity of the color are the issues. Other formulations produce non-biodegradable or semi-toxic waste products. These must be properly treated and made harmless prior to returning the treated water to the environment. These concerns exist worldwide.
Fiber Forms Used For Dyeing
Dyes are specific to the fiber involved and this factor will be discussed in some detail later in this document. However, they are not specific to the type of substrate the fiber is used. For example, the dyeing process can be accomplished using textile fibers, yarns, fabrics, or garments. All of these different textile substrates can use the same dyes as long as the fiber content is the same. However, there are reasons for dyeing different fiber forms.
- Fiber dyeing is used as a styling technique. Natural fibers or staple synthetics are dyed in bundles or baskets. This process is known as ‘stock dyeing’. These fibers are often blended for shade in the yarn manufacturing process. They are used to produce heather shades which are popular in many different apparel products.
- ‘Dope or solution dyeing’ is the process where color is mixed into the polymer solution prior to fiber extrusion. This process yields colored fiber with very high fastness properties. However, it is difficult to alter the extruded color if off shade. Certain synthetic fibers such as polyethylene can only be colored using this technique. These fibers are used where high color fastness is a must, such as geotextiles. In general, fiber dyeing is inflexible with regard to color flexibility. It is also a more expensive technique because large amounts of colored fiber waste are generated in the manufacturing process.
- Yarn dyeing is also used as a styling technique. Yarn dyeing is used to produce stripes, plaids, and some complex designs with 100% fiber content products. In many cases, the same designs could be obtained by textile printing. However, yarn dyeing produces products with higher color quality and color durability. ‘Skein dyeing’ is the process where a single yarn is wound into long yardage loops or hanks. The hanks or skeins are held loosely in the dyeing machine. The dyebath cascades like a waterfall over the loops of yarn. This allows the yarn to develop more volume or bulk during the dyeing process. This technique is important for products like rugs or sweaters.
- In ‘package dyeing’, a single yarn is wound uniformly around a perforated tube or spool. Multiple of these packages are loaded into the dyeing vessel by placing them onto perforated carrier arms. The dyebath is pumped through the package inside-out and outside-in until the dyeing process is completed. This is the most versatile and highest productivity method for dyeing yarns. Yarns dyed in this way are used in a wide variety of products.
- ‘Beam dyeing’ is a technique where multiple yarns are wound side by side onto a single perforated beam. This can be a few hundred or even a few thousand yarns wound onto a single beam. This is dependent on the end product and manufacturing requirements. ‘Beam dyed’ yarn is often used for striped or plaid woven fabrics. Typically a single beam is placed in the dyeing vessel and the dyebath is pumped through the yarns in the inside-out and outside-in flow technique.
- ‘Space dyeing’ is a yarn dyeing method for placing color sections or blocks along the length of the yarn. The most popular method is a modification of package dyeing and will be discussed in more detail later in this document. Space dyeing is a highly specialized coloration technique. These yarns are often used for highly styled sweaters. Synthetic fibers dyed this way are used in the carpet industry. In general, yarn dyeing is more expensive than fabric dyeing due to lower productivity and colored yarn waste.
- Fabric or piece dyeing is the most cost-efficient and highest productivity technique. Fabric dyeing machines include jet machines, dye becks, fabric beams, and jig dyeing machines. Each type of machine has its own unique advantages and limitations. These will be discussed later. All of these machines as well as most of the yarn dyeing machines are batch or exhaust machines. Fabrics can also be dyed on continuous pad dyeing ranges or using the semi-continuous pad batch method. Textile printing is a localized continuous dyeing technique. Fabric dyeing produces the largest amount of dyed textile materials. However, there is colored cut and trim waste produced when cutting and sewing dyed fabrics into garments.
- Garment dyeing is a batch dyeing process and is very popular with certain types of products such as sweaters or hosiery. This method uses commercial laundry machines such as a rotary drum or paddle machine to dye garments. It is a slower, lower, productive, less efficient method than fabric dyeing. However, garment dyeing produces no colored waste and is the most color-responsive technique to the demands of the marketplace.
Factors in Exhaust Dyeing
The vast majority of textile substrates are dyed using the exhaust or batch method. Figure 14 illustrates the general process of batch or exhaustion dyeing.
The dyebath consists of water, dyes, and any necessary auxiliary chemicals. The textile substrate is immersed in the dyebath. The dyebath is heated to the dyeing temperature and held at that temperature for the required amount of time. Simultaneously, the fabric is agitated during the process. During the process, the dye migrates and ‘adsorbs’ to the surface of the fiber. Subsequently, the dye molecules ‘absorb’ and diffuse into the fiber structure. This process is known as exhaustion. Once all possible dye diffuses into the fiber, residual dye is removed by after-washing.

The actual color of the textile material obtained from this process is determined by the amount or weight of dye in the dyebath formulation. This is usually expressed as a percentage of the weight of the textile material being dyed and is known as ‘depth of shade’. Also, for most commercial dyes, only a fraction of the total dye in the bath actually exhausts into the fiber. This is known as ‘fractional exhaustion’ and is a key parameter of each dye in the formulation. The higher the fractional exhaustion, the better the use of the dye and the less dye or color in the wastewater.
Other key factors affecting exhaustion dyeing include substrate preparation, machine agitation, flow characteristics, and ‘liquor-to-goods ratio’ (LR). LR is a machine-based parameter defined as the weight of the dyebath compared to the weight of the textile substrate. Of course, with any textile dyeing process temperature, pH, chemical auxiliaries, and time of dyeing are key control factors. Other factors that influence the dyeing performance are related to the specific dyes and/or specific fibers used in the process. It is essential to have properly cleaned machines to prevent dye spots or cross-staining contamination from dye lot to dye lot.
Dyes for Cotton (Cellulose) and Blend Fibers
As mentioned earlier, dyes are fiber specific based on the chemical nature of the dye and the molecular nature of the fiber. Table 3 is a chart that shows the most commonly used textile fibers and what classes of dyes work with each fiber.
| Type of Fiber | AcidDye | Basic Dye | Direct Dyes | Disperse Dyes | ReactiveDyes | Sulfur Dyes | Vat Dyes |
|---|---|---|---|---|---|---|---|
| Natural | |||||||
| Cotton | X | X | X | X | |||
| Wool | X | X | |||||
| Synthetic | |||||||
| Rayon | X | X | X | X | X | X | |
| Acetate | X | ||||||
| Polyester | X | X | |||||
| Nylon | X | X | |||||
| Acrylic | X | X | X |
Table 3 – Comparison of Fibers and Dyestuffs Used
Dye classes are actually groups of dyes that work on a specific textile fiber in a particular way. These dyes within a group will have similar chemical compositions but they will not be identical. Dye classes can be thought of as application groups of dyes.
From Table 3, the dyes that work for cotton are reactive, directs, vats, and sulfurs. Naphthol dyes are not used on cotton and rayon as much as in previous years. Pigment colorant properties will also be discussed. However, pigments are not dyes and they are not fiber specific. All the dyes used for cotton will also work for other natural cellulose fibers such as linen or ramie. They will also work for the synthetic cellulose fibers such as rayon, lyocell, and bamboo. As mentioned earlier, the final color of any specific dye formulation on cotton will be different than the color on rayon, even if the fibers are dyed together.
Currently, reactive dyes are the most used for cotton blend fabric. These dyes react directly with the chemical structure of the cellulose. They form a strong covalent bond between the dye and the fiber resulting in very good to excellent wash fastness. With proper after washing, these dyes also exhibit good crock fastness. Their resistance to sunlight is variable, but most reactive dyes have acceptable lightfastness.
Reactive dyes were invented in 1956. They have the widest shade range with the brightest colors that are currently available for cotton. They are highly water-soluble and are useful for both exhaust and continuous dyeing. There are many different types of chemistries used for reactive. The cold dyeing type can dye cotton at low temperatures. These dyes are used in the cold pad-batch energy-saving dyeing technique. The hot dyeing type is more difficult to exhaust into the fiber requiring higher dyebath temperatures for long periods of time. Many new reactive dyes can react at two positions on the fiber. These ‘bi-reactive dyes’ exhibit superior fastness properties but are more difficult to process. They typically require higher dyeing temperatures and longer processing.
Unfortunately, some of the reactive dye in the bath can react with the water under dyeing conditions and become deactivated. The deactivated dye cannot be recovered and becomes a colored wastewater pollutant. Normally, reactive dyes typically require large amounts of salt, such as sodium sulfate as a dyeing assistant. This salt can be an environmental concern in wastewater. Reactive dyes are not generally chlorine resistant; however, it is dye dependent. These dyes require large volumes of water for the processing which can be a negative cost factor. They are moderately expensive dyes; however, specific dye costs per pound can vary greatly. In general, newer patented dyes are more expensive than older generic dyes. Overall, reactive dyes are the cotton dyestuff of choice for most end uses. Their combination of a wide range of bright colors and good fastness properties, especially wash fastness, make them the best dye choice for most cotton products.
Direct dyes are a grouping of dye structures that have been used for many years with cotton substrates. They are highly water-soluble and used mainly in exhaust dyeing. They are usually temperature-sensitive with most being dyed at temperatures between 160-180oF. Like reactive dyes, they use salt as a dyeing assistant but need very small amounts compared to reactive dyes. They do not react with the fiber structure but rely on dye-to-fiber associations such as hydrogen-bonding and dipole moments to help with color fastness after dyeing.
As a group, they have a wide shade with fairly bright colors, but not as bright as reactive dyes. They are easily exhausted during dyeing. In general, they exhibit good, light fastness. However, they have poor wash fastness and poor wet crock fastness. Direct dyed medium and dark shades tend to bleed and fade in standard home laundry washing. Chemical after-dyeing treatments known as dye fixative agents (dye fixes) have been developed for direct dyes. Some of these have greatly improved the wash fastness of directs but can change the shade of the dyeing. In general, direct dyes are not a good choice for items that will be washed frequently. However, they are a good choice for items such as drapes or curtains where lightfastness is the major requirement. Finally, they use much less water, chemistry, and time compared to reactive dyeing.
Vat dyes are a group of older dye structures. These dyes are not water-soluble in their normal form. In order to dye cotton, these dyes are made water-soluble by the chemical process known as ‘reduction’. This requires a reducing agent such as sodium hydrosulfite and an alkali, typically sodium hydroxide. The reduced form of the dye is known as the ‘leuco form’. For most vat dyes the leuco form is highly water-soluble and nearly colorless or very lightly colored. In the leuco form, the dye is highly attracted to the cotton fiber. It rapidly absorbs into the fiber structure. Once inside the fiber, the leuco form of the dye is ‘oxidized’ back to the water-insoluble original colored dye.
This process traps a water-insoluble dye inside the cotton fiber. The dyes are high wash fastness and usually very lightfast. With proper after washing they exhibit good to excellent wet and dry crock fastness. Most of the vat dye structures are highly resistant to chlorine. As a group, these dyes exhibit the best color fastness properties on cotton fiber.
Unfortunately, vat dyes are limited in their shade selection. There are very few yellow, orange, or red dyes available and these tend to be very dull. There are relatively bright dyes available in deeper colors, particularly blues, greens, and blacks. However, vat dyes produce overall duller shades than reactive or direct dyes. The dyeing procedure is cumbersome involving several complex chemical steps. Control of these conditions determines whether the dyeing process is successful or not.
Additionally, the vat dyes are by far the most expensive dyes used for cotton on a cost-per-pound basis. Due to the complex dyeing process, vat dyes are used more with continuous dyeing than in exhaust processing. Vat dyes are a good choice where color fastness especially wash fastness is a prime requirement, but where a wide shade range of colors may not be as important. Vat dyes are typically used for work wear, towels, and high-end garments which must survive multiple items of washing such as men’s dress shirts and dress pants.
Indigo is an ancient natural dye that happens to be a vat dye. Today, synthetic indigo is the dye mostly used by the textile industry. When dyed onto the cotton fiber, indigo creates a bright reddish blue shade. Indigo is used for denim and is, by volume, the most used dye in the textile industry. Indigo forms a ring of dye around the outer edges of the fiber. Figure 15 shows the ring dyeing nature of indigo.

Since the color ring of indigo is around the outer fiber surface or the outer surface of the yarn bundle, indigo leaves a white inner core. This dyeing can be abraded in garment finishing (stone washing, hand sanding, laser, etc.) to produce special color effects. Additionally, unlike other vat dyes, indigo can be destroyed or discharged with sodium hypochlorite bleach or potassium permanganate treatments. These special processes are performed on denim garments and are possible due to the chemical sensitivity and ring dyeing nature of indigo. Figure 16 illustrates a schematic of an indigo yarn dye range.

Denim is a yarn-dyed fabric where only the warp yarns are dyed. A ball warp is a package of 400 yarns made into a rope. A typical dye range dyes 10-36 ropes of yarn simultaneously. The ropes are fed into the preparation section of the range where detergent and alkali help clean impurities from the yarns so that the indigo can penetrate and dye the yarns more uniformly. As an option sulfur black dye can be applied (sulfur bottom) in this section to make the indigo dyed yarn appear deeper in color. Indigo does not exhaust in normal processing.
To obtain deep blue color, layers of indigo are built up in ring form. This is done by dipping the yarn in the reduced (leuco) indigo dyebath and then oxidizing this layer of dye by exposing the leuco indigo to oxygen found in the air (skying). In this way, layer upon layer of indigo is built on the yarn and fiber.
Typically, 6-8 dips and skyings are required to produce standard denim color depths. In the final section of the range, an optional sulfur top of sulfur black dye can be applied to make the shade appear to be a little blacker. However, in standard processing, the final section of the range is only used for washing to remove unfixed, surface indigo.
Any indigo removed can be captured and reused. The final washing of the dyed yarns helps minimize color transfer during weaving and crocking problems in the final fabric. The dyed and washed yarns are then dried and segregated into individual ropes. The yarns of each rope will be separated and wound onto section beams, ready for the weaving process. There are many optional processes in producing modern denim products but the nature of indigo is still the key factor. Unlike other vat dyes, indigo is an average-priced dye.
Sulfur dyes are the least costly dyes available for cotton. Like vat dyes, in their normal form, sulfur dyes are water-insoluble. Also, like vats, they are reduced to a water-soluble form, applied to the cotton fiber, then oxidized back to their water-insoluble form once the dye has been absorbed into the fiber structure. Unlike vats, the reduced water-soluble form of the sulfur dye does not change color. Sulfur dyes have a low color power so a large amount of dye is necessary to produce deep shades. Sulfur dyes can be applied in either exhaust or continuous dyeing techniques. When properly dyed and after washed, these dyes exhibit good wash fastness. However, their crock fastness is limited, especially for deep shades.
Sulfur dyes have a fairly wide shade range but they produce dull colors. In light shades, these dyes tend to have poor lightfastness. However, in dark shades, these dyes exhibit good lightfastness so they are often used for navy, blacks, and greens. In storage, particularly hot, humid conditions, sulfur-dyed products can form sulfurous and sulfuric acids which attack and destroy cellulose. Sulfur-dyed products must be finished in a manner to neutralize this acid formation. Otherwise, severe fabric strength loss will result from storage. Sulfur dyes are a good, inexpensive choice, particularly in deep shades, where wash fastness is important but shade brightness is not a major requirement. In woven bottom weights, sulfur black is the standard due to its rich shade.
Naphthol dyes are an older technology dye system. With this dyestuff class, a water-insoluble dye is created inside the fiber during the dyeing process. Basically, a component known as a naphthol is applied to the cotton material. The naphthol component is not a dye and can be washed out of the substrate. Next, a second component known as a coupling salt is joined to the naphthol during a process similar to exhaust dyeing. The color salt reacts with the naphthol to form a water-insoluble dye inside the fiber. The technology was developed to produce bright yellow, orange and red shades with vat dye fastness properties. However, this process is very complex. It is also difficult to shade match. These dyes have lost nearly all of their market share to reactive dyes. However, some naphthol dyes may still be used for some specialty products and in textile printing.
Pigments are totally water-insoluble colorants. They have no affinity for textile fibers and must be attached to the fiber or yarn bundle surface with a binder or glue. The binders are not fiber specific and can be used on different fiber types simultaneously. Pigments do not penetrate into the fiber structure. In general, pigment coloration exhibits good lightfastness. Their washfastness properties are directly dependent on the binder selection.
Typically, they produce intense colors with a wide shade range. They are simple to use. Pigments are useful in printing, continuous pad or spray coloration. Some specialty types, known as exhaustible pigments, can be used in garment dyeing. They usually produce colored garments with distressed or garment washed appearance without using an extra garment washing process. However, they have become more expensive and sometimes stain the processing equipment. Many pigments are fluorescent and are used to produce ‘neon’ colors. Pigments typically are lower cost colorants than dyes.
Since the colorants are glued to the outer surface of the yarn in the fabric, pigments have limited crockfastness, especially dry crockfastness. The binder systems tend to make the feel or hand of the textile feel stiff or harsh. In general, the better the pigment fastness properties, the stronger the binder and the stiffer the hand. When applied by padding or spraying, pigments can migrate during drying. This leads to uneven color or even blotches in the colored product. During processing, the binder may cause build- up on machinery rolls or may cause sticking of the substrate.
Pigments are also economical because of the limited number of processing steps. Blends can be colored to a solid or union shade with one pigment formulation which is applicable to all fibers. Pigments offer the possibility of combining coloration and finishing. However, the specific pigments and finishes must be chosen so that their normal processing conditions match.
Disperse dyes are water insoluble dyes which are used for thermoplastic synthetic fibers, primarily polyester. They are only medium quality dyes for acrylic fibers and they are generally poor dyes for nylon. Disperse dyes are never water soluble. They are dispersed in particulate form during the polyester dyeing process.
During processing, the polyester fiber is swollen by either a chemical swelling agent known as a carrier or by high temperature in the range of 265-275oF. Carrier dyeing is processed with water up to a boil (212oF). In order to get water to those temperatures a high temperatures pressurized dyeing vessel is required such as a jet machine for fabric dyeing or a package machine for yarn dyeing. Once the proper conditions are reached, the disperse dye diffuses throughout the polyester structure. After cool down, the polyester substrate is afterwashed to remove unfixed dye. Disperse dyes exhibit good washfastness, light fastness and crockfastness. They are typically used for all cotton/polyester blends.
Disperse dyes are an average priced dyes. They do not dye cotton fibers but may stain them. This staining is easily removed during processing, but may require an additional step. Some disperse dyes have the capability of sublimation. Sublimation is the property where upon severe heating, the solid dye particle changes directly into a gas, without going into the liquid or melted state. The dyes then immediately revert back to the solid state under proper temperature conditions. Disperse dyes that sublimate are used in the continuous dyeing process known as Thermosol dyeing and in sublimation printing. Disperse dyes are the only textile dye class that can sublime.
Acid dyes are used in the dyeing of nylon, wool and silk. They are highly water soluble. Under dyeing conditions, these fibers generate a positively charged dye site on the fiber structure. At the same time, the acid dye develops a negative charge. This negative- positive charge attraction is the initial driving force to move the dye from the dye bath into the fiber structure. Once inside the fibers, other dye-fiber interactions account for the final overall colorfastness of the dyed fiber.
There is a wide range of acid dyes with a wide color range and a wide variety of colorfastness properties. However, acid dyes are the dyestuff of choice for use with cotton-nylon blends. In general acid dyes do not dye cotton; however, staining can be a problem. This must be evaluated on a dye formulation by dye formulation basis. Any staining should be removed during processing.
Basic dyes are water soluble dyes used for dyeing acrylic fibers. Under dyeing conditions, the acrylic fiber generates negatively charged dye sites while the dye generates a positive charge. The positive-negative electronic attraction is the initial driving force to move the dye from the dyebath into the fiber. Once inside the fiber, other dye-fiber interactions influence the final colorfastness properties of the dyeing. Basic dyes have a wide color gamut and exhibit a wide range of colorfastness properties. Several of these dyes are fluorescent (neon dyes) or produce extremely bright colors. These ultimate brightness dyes tend to have poor lightfastness. Basic dyes are the dyestuff of choice for acrylic fibers and are used with cotton-acrylic blends. They can produce severe staining on cotton and this must be taken into account when dyeing these blends.
There is always a range of properties for specific dyes within any dye class. Also colorfastness properties, in particular, can vary depending on the specific fiber content of the dyed textile substrate. For example, reactive dyes have better washfastness properties on cotton substrates than they do on rayon substrates. However, reactive dyes are a good dye choice for both types of fibers. There are many other examples of this situation.
Dyeing Blends
A large portion of the dyed textile substrates are actually fiber blends. Worldwide, the most popular blend is cotton and polyester. Other fiber blends are also very popular. Of particular interest are the cotton and spandex blends. In the dyeing process, all fibers in the blended product must be considered individually. Using a cotton/polyester blend as an example, the cotton would be dyed with reactive dyes while the polyester portion is dyed with disperse dyes. The dyeing procedure is also modified to accommodate each fiber in the blend. For a cotton/polyester substrate, the polyester portion is usually dyed first followed the dyeing of the cotton.
The reason for this sequence is that the conditions necessary for dyeing polyester could damage dyed cotton fibers. However, the conditions needed to dye cotton have little or no effect on the dyed polyester fibers. Also dyeing times required for blends are typically much longer than the times needed for 100% fiber content substrates.
In practice, spandex does not dye but it will stain with various dyes. In many applications of spandex, it is normally covered with another dyeable fiber such as in core spun and cover yarns. In other products, the individual spandex filament is hidden on the back of the fabric. Normally, the end product requires that the spandex be stained enough to not be noticed. However, spandex colorfastness, particularly washfastness, is not good and this can present some quality issues in end products.
When different fibers in the blend are dyed different colors, this process is known as ‘cross dyeing’. Because of the fiber specific nature of dyes, this is a relatively easy dyeing process. For instance, out of a single dyebath disperse dyes will only dye the polyester and direct dyes will only dye the cotton. If reactive dyes are used, separate dyebaths will be used for the polyester and the cotton.
‘Union dyeing’ is the term used when the blended substrate is dyed to a uniform, solid color. A true union requires that each fiber in the blend be the same color. This can be very difficult and requires a high degree of skill on the part of the dyer. Using our cotton/polyester example, the dye formulation of disperse dyes on polyester will have to produce the same shade as the reactive dye formulation produces on the cotton fibers. One of the biggest problems with this is that the two fibers have a different brightness or luster. Regardless of the difficulty, high quality union dyeings are produced every day on blended substrates.
When at least one fiber in the blend is left undyed, the term ‘reserve dyeing’ is used. If the blended substrates are intimate blends, then reserve dyeing produces a heather appearance. In our example, the cotton fibers could be dyed with reactive dyes and the polyester left undyed. Alternatively, the polyester fibers could be dyed with disperse dyes and the cotton left undyed. Depending on the blend ratio of cotton to polyester, these two reserve dyeings could look very different when compared to each other. Also, staining of the undyed fiber may or may not be removed depending on the end product requirements.
When different fibers which dye with the same dye classes are blended together, a tone-on-tone effect results from the dyeing process. For example, cotton and rayon can be blended together. They both dye with reactive dyes. After dyeing both fibers will be the same hue or color, however, the rayon fibers will be a deeper shade than the cotton fibers. This is due to the fiber structural differences between cotton and rayon. Normally dye penetrates rayon more easily then cotton resulting in the deeper shade.
When dyeing blends, the properties of the weakest fiber in the blend must be considered. The standard dyeing method for one fiber may severely damage another fiber. Therefore, in many cases, certain dye formulations, chemical auxiliaries and dye bath conditions which are considered routine will be modified or eliminated due to the sensitive nature of the blend fiber.
Dyeing Equipments
The purpose of dyeing equipment is to contain the textile substrate and the dyebath and to generate an exchange between the substrate and the dye. It also must monitor and control of all necessary dyebath parameters including temperature levels and degree of agitation. Most dyeing equipment is made of stainless steel in order to resist chemical attack and corrosion from the dyebath chemicals at the elevated temperatures required for most dyeing processes. Most dyeing machinery control systems incorporate microprocessors or computers. This has greatly improved the accuracy, monitoring, flexibility, and reproducibility of the control systems.
Most dyeing equipment is designed to process specific textile substrates. That is, there is dyeing equipment designed to dye yarns. These are not used to dye fabrics. There are machines designed for dyeing fabrics. These are not used to dye yarns. There are also machines designed for dyeing garments. These machines are not used for dyeing yarns, but can do limited amounts of fabrics.
Most dyeing equipment is designed for exhaust or batch dyeing. These machines are targeted to use with yarns or fabrics or garments. There are continuous dye ranges designed for dyeing fabrics. Pad-batch is a semi-continuous dyeing range designed for dyeing fabrics. The indigo dye range described earlier is a continuous yarn dyeing range.
Yarn Dyeing Machines
Figure 17 illustrates a package yarn dyeing machine. A single yarn is wrapped around a perforated dye tube to form a package. The size of the package is based on the weight of the yarn wound onto the package. Typical package weights used in most package machines today are 3 to 5 lbs. The capacity of the dyeing machine is expressed as a total yarn weight that can be dyed in a single dyeing. For example, one machine may be rated at 400 lbs., while a larger machine is rated at 1,000 lbs.
Package machines are batch, exhaust dyeing machines. The correct number of yarn packages is loaded onto perforated carrier tubes. The carrier is placed in the machine. The vessel has a lid which is sealed. Most package machines can be pressurized in order to dye polyester. During the process, the dyebath is pumped through the package with inside-out flow, then the flow is reversed to outside-in. This is done to insure color uniformity throughout the package.
Once dyed, the packages are washed and removed from the machine. They are then dried. The quality of the dyeing is evaluated by back-winding yarn from the inside, middle and outside of the package. This back-wound yarn is then knitted into a sleeve, and color uniformity as well as shade are evaluated. Typically, 3-7 packages will be evaluated per dye lot. Package dyeing is the highest production, most widely used method for dyeing yarn.
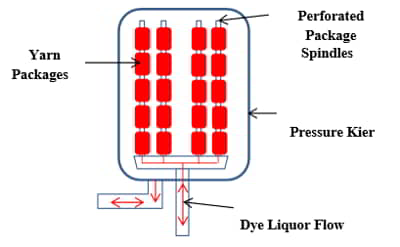
Space dyeing is a specialty yarn dyeing method where variable segments of different colors are dyed along the length of the yarn. Space dyeing is a semi-continuous technique that is a modification of package dyeing. In this method, a package of yarn is wound onto a dye tube that is placed in the space dyeing machine. Dye is then injected into specific quadrants of the package using needles which are inserted into the package. Each needle injects a different color and inserted to different depths. The typical number of needles used is 4 or 8, but more or less can be used.
Once the dye is injected, the needles are removed and the packages are steamed to fix the dye. The fiber type and dyes used must be those that can be fixed with steam. The dyed packages are then loaded into a package dyeing machine and afterwashed, and then unloaded and dried. The yarn is then back wound onto cones for further processing. Space dyeing produces unique coloring effects that cannot be obtained on other dyeing equipment. Figure 18 shows the needle assembly used and a package of space dyed yarn. Figure19 shows a sweater with space dyed yarns.


Fabric Dyeing Machines
Most textile products in the market today are dyed in fabric form. The most popular type of dyeing machines for fabrics is the ‘jet dyeing machine’. Figure 20 shows a schematic of a typical jet machine.
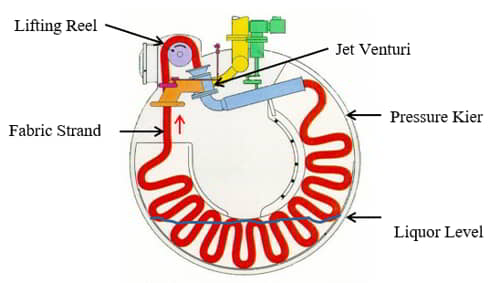
The fabric is loaded into the machine in rope form. The strand of fabric travels around the lifter reel at the top of the machine, then through the jet nozzle tube (venture), and onto the back of the machine. The cloth, typically 300-400 yards, is sewn into a continuous loop, and travels around the machine continually during the process. The length and weight of the fabric making up a continuous loop will be determined by the machine capacity. Many machines can dye many roped strands totally several thousands of pounds of fabric in a single dyeing.
As the fabric circulates within the machine, the dyebath is constantly pumped from the bottom of the machine through a filter and then through the jet nozzle. This venture provides the force to move the rope of fabric. Therefore, during the dyeing process, the dyebath is circulated and contacts the fabric in the venture tube, resulting in excellent agitation and high efficiency dyeing.
After dyeing, the fabric is afterwashed, unloaded and dried. Jet machines are sealed, pressurized batch, exhaust machines. Normally, they are computer-controlled and considered the most flexible machinery design for dyeing all types of fabrics, knits and wovens. The jet machine was invented in the 1960’s but is the dominant fabric dyeing machine today.
Jet machines are manufactured by various companies throughout the world. In many cases, new machines maintain the basic jet machine operations, but their designs are modified to meet specific textile fabric requirements. Figure 21 illustrates a horizontal- flow jet that can be compared to the conventional jet configuration in Figure 20.
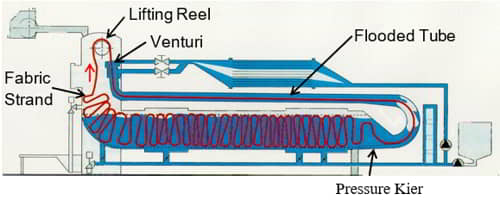
The “dye beck” is one of the oldest types of dyeing machines used for fabric dyeing. The basic design for the dye beck was developed in Europe in the 1500’s. Until the invention of the jet dyeing machine in the 1960’s, the dye beck was the most widely used exhaust fabric dyeing equipment. Figure 22 is a schematic of the design of a typical dye beck.

Like the jet, the fabric is loaded into the machine in rope form over an idler reel at the front and around an elliptical reel at the back of the machine. The dyebath is held in the bottom of the machine. The typical cloth strand length is approximately 100 yards but this can vary with machine capacity. Production machines typically are rated from a few hundred to many thousand pound capacity using numerous strands of fabric.
During the dyeing process the fabric is pulled around the machine by the rotation of the elliptical reel. The dyebath remains stationary. This agitation creates stress on the fabrics during the process and tends to stretch and distort knit fabric structures. After dyeing the fabric is afterwashed and the cloth unloaded. Dye becks are still widely used. They are simple to operate. Some newer designs are usable with knits. In general, dye becks use much more water and energy and generate more wastewater than the jet machines.
The ‘fabric beam dyeing’ machine is a batch, exhaust machine designed to process surface sensitive or easily distortable fabric such as lightweight warp knits and power stretch fabrics of all types with cotton/spandex blends. Figure 23 is a photograph of a typical fabric beam machine.

Using this machine, fabric is wound open-width around a perforated beam. The beam is loaded into the machine and the door is sealed. During dyeing, the dye is pumped through the cloth in an inside-out flow. In this process the dye bath is circulated while the fabric is held stationary. This minimizes stress and distortion of the fabric during dyeing. The fabric can be dyed essentially wrinkle-free. After dyeing, the fabric is afterwashed, then unloaded and dried. The machine shown in the figure is sealed and pressurized in order to dye the polyester in cotton-polyester blends at high temperatures.
Overall, fabric beam dyeing machines can handle up to 3,000 pounds. They are considered to some degree specialized dyeing machines. Figure 24 is a schematic of the flow characteristics of a beam dyeing machine.
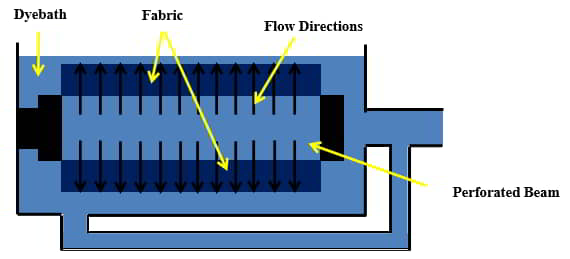
‘Continuous dye ranges’ are high production primarily for woven fabrics but are highly expensive dyeing equipment. Figure 25 shows a schematic of a standard dye range. Continuous dye ranges are actually a series of different machines that control certain dyeing conditions as the fabric is processed. From Figure 25, the open-width fabric is saturated with concentrated dye liquor, and then squeezed through pad rolls to remove any excess dye. Very accurate dyeings can be obtained by the ability to control the concentration and the amount of dye applied during padding. The fabric is then passed through a pre-drier to minimize dye migration. The fabric is then dried to a specified moisture level, and then passed through a chemical pad to apply the correct amount of auxiliary chemicals necessary for the dyeing process. In the case of reactive dyes on cotton fabric, salt and alkali are applied in the chemical pad. The fabric immediately goes into the steamer and dwells in this steam atmosphere long enough to fix the dye. Steamer dwell times for most dyeings vary from 10-30 minutes. After fixation, the fabric is passed through a series of washers to remove unfixed dye. After the washing process, the fabric is usually dried before further processing.
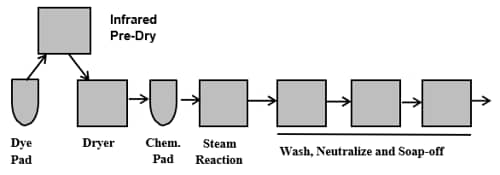
Continuous dye ranges are complex operations. They require large amounts of floor space and are very expensive to purchase, install and maintain. They require more down time than other dyeing machines for cleaning or maintenance. However, this is offset due to the large amounts of fabric that can be dyed in a short amount of time. Normal production speeds are up to a slightly above 100 ypm. Another big advantage is they process the cloth flat and wrinkle free. Due to the high level of linear stress on the fabric, they are targeted toward wovens. As a general rule, they are shade inflexible in that they are only profitable for long runs of the same shade. In recent years, they have lost some market share to exhaust dyeing.
Today, energy, in the form of electricity, steam, and natural or propane gas, is a major cost item in the dyeing of textile substrates. Because these energy concerns, a ‘pad batch’ dyeing of cotton fabrics has become more common place. This system uses the specialized properties of certain reactive dyes. This new semi-continuous method is known as pad-batch dyeing and is illustrated in Figure 26.

In pad-batch dyeing, flat open-width cotton fabric is saturated with selected concentrated reactive dyes, squeezed through a pad and rolled up. The dye saturated fabric is then loaded onto a motorized A-frame. The cloth roll is covered with plastic to prevent surface evaporation and the roll is slowly rotated or batched to prevent gravity from moving the dyestuff and insuring uniform dyeing. The batch time can be from 4 to 24 hours depending of the type of reactive dye used. This entire process is at room temperature requiring very little energy. During the batching process, these special cold reacting reactive dyes fix onto the cotton fabric. Most hot dyeing reactive dyes will not work well with this process. After fixation, the dyed fabric is washed to remove unfixed dye and the fabric is dried.
Pad-batch dyeing is a simple, low energy method for dyeing cotton fabrics. It only works with certain reactive dyes so there may be limitations in shade availability and fastness properties of the dyed fabric. This is an extremely energy efficient method capable of high productivity requiring a small amount of plant floor space. It is currently used worldwide for cotton fabrics.
‘Garment dyeing’ has become popular for certain types of garments such as sweaters and socks. In addition, knit products such as t-shirts and knitted pants are dyed with this method. Recently, denim garments which have been stonewashed or discharged are over-dyed in garment form. Figure 27 is a photograph of the most popular type of machine for garment dyeing known as a rotary drum machine.
The rotary drum machine used for garment dyeing had its roots in the commercial laundry industry. These are batch, exhaust dyeing machines. They have a rotating basket inside the machine into which garments are loaded. As the basket rotates, the garments tumble creating agitation with the dye bath which is necessary for dyeing. The tumbling action can be modified depending on the internal design of the basket. In general, dyeing procedures are the same as with other exhaust machines. After dyeing, the garments are washed. Some modern machines have garment extraction spin cycles. The garments are unloaded and transported to further processing.

The justification for garment dyeing has been discussed earlier. However, this method is lower productivity than fabric dyeing but produces no colored waste fabric since only the fabric that is sewn into the garment is dyed. Also, since the dyeing is closer to the retail industry in time (after cut and sew) the dyeing of unpopular colors can be minimized. This method is most effective for cotton knit structures; however, woven garments like denim are over dyed. Typically, exhaustible pigments are processed using this type of machine. Some rotary drum machines can be pressurized and are used for dyeing cotton-polyester blends. A big advantage of garment dyeing is that process time is reduced due to much less water being used and faster after washing.
Summary of Dyeing
The dyeing process, regardless of the textile substrate, is complex and requires a high degree of knowledge and skill on the part of the manufacturers. Dyeing requires clean textile substrates in order to maximize product quality. In preparation, desizing and scouring are required processes for woven fabric structures. Other textile substrates only require scouring. Scouring and bleaching are necessary for whites, pastels and very bright shades. Most other preparation processes are optional. Clean process water is critical for producing quality products.
Dyeing can be complex and even cumbersome. Dyeing quality and performance are directly controlled by time, temperature, pH, and chemical auxiliaries. Dyes are fiber specific and have their individual advantages and disadvantages. They must be processed under careful control. What may be thought of as an insignificant variable can have a major impact on dyeing performance and dyed product quality. Dyeing equipment is designed for specific types of textile substrates and, in certain cases, specific dyeing conditions.
The goal of dyeing is to produce the highest quality on-shade product which meets all of the customer’s specification for colorfastness and color durability. This dyed product should be produced in the shortest amount of time, using the least amount of energy, generating minimum water waste. This produces the most profit for the manufacturer and the highest quality product for the customer.
References
- Hudson, P., Joseph’s Introductory Science. 6th Orlando, Fl.: Harcount, Brace, Jovanovich; 1993.
- Berns, Bill Meyer and Saltzman’s, Principles of Color Technology. 3rd Edition, New York, NY; John Wiley & Sons, Inc.; 2000.
- Dyeing Primer, Research Triangle Park, NC: American Association of Textile Chemists and Colorists; 1981.
- Hatch, K, Textile Science, Los Angeles, CA: West Publishing Company,









Howdy! I know this is kind of off topic but I was wondering which blog platform are you using for this site? I’m getting sick and tired of WordPress because I’ve had problems with hackers and I’m looking at options for another platform. I would be great if you could point me in the direction of a good platform.